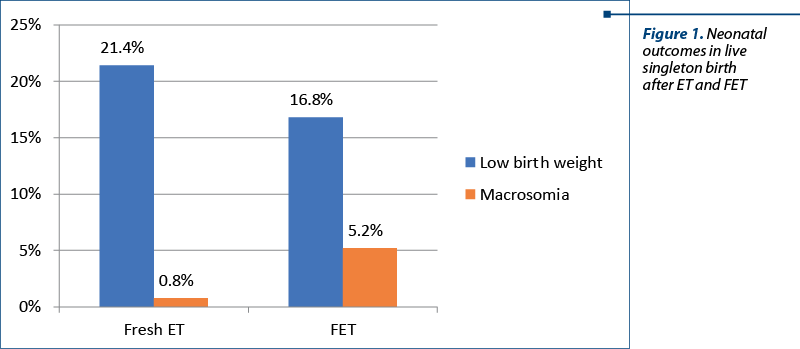
Purpose. To assess if the frozen-thawed embryo transfer (FET) results in better birth outcomes than fresh embryo transfer (ET). Methodology. Our retrospective study involved women undergoing in vitro fertilization (IVF) between 2017 and 2019 using autologous eggs and having a singleton pregnancy delivered from the 37th week onward at an IVF centre in Târgu-Mureş. The study included 121 and 95 patients with fresh and frozen embryo transfer, respectively. Furthermore, patients with normal BMI, non-smokers and aged under 40 years old were included. We compared the rates of mean birth weight, low birth weight (LBW <2.5 kg) and high birth weight (HBW>4 kg), after fresh versus frozen embryo transfer in normogonadotropic healthy young women undergoing IVF. Results. The mean neonatal birth weight was higher after FET than after ET (3316 g FET versus 3154 g ET). The result of the trial suggested that the FET protocol was associated with significantly lower rates of LBW (16.8% FET versus 21.4% ET; p<0.05) and with higher rates of macrosomia (5.2% FET versus 0.8% ET; p<0.03) than the fresh ET protocol. The assessment of 216 babies showed no statistically significant difference in the mode of delivery (C-section versus vaginal birth) and sex of live born between the two study groups. Conclusions. The risk of high birth weight following frozen/thawed embryo transfer was greater than that following fresh embryo transfer, but the risk of low birth weight among frozen/thawed embryo transfer neonates was significantly decreased in comparison with fresh embryo transfer neonates.
Caracteristicile neonatale după transfer de embrioni fresh sau congelaţi la paciente normogonadotrope, tinere şi sănătoase
Neonatal outcome after fresh versus frozen embryo transfer in normogonadotropic healthy young women undergoing IVF
First published: 18 mai 2020
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.28.2.2020.3182
Abstract
Rezumat
Scop. A evalua dacă transferul de embrioni congelaţi (FET) are efecte benefice asupra greutăţii nou-născutului, comparativ cu transferul de embrioni fresh (ET). Metodologie. Studiul nostru retrospectiv a implicat femei care au avut fertilizare in vitro (FIV) între 2017 şi 2019, cu ovocite proprii, în Clinica de fertilizare Zygota din Târgu-Mureş. Criterii de includere: paciente cu sarcină monofetală, care au născut la termen (după săptămâna 37 de gestaţie), cu indice de masă corporală normal, nefumătoare şi cu vârsta sub 40 de ani. Studiul a inclus 121 de sarcini unice după ET şi 95 de sarcini unice după FET. Am comparat ratele de greutate medie la naştere, greutate mică la naştere (<2,5 kg) şi greutate mare la naştere (>4 kg), după ET sau FET, la paciente normogonadotrope, tinere şi sănătoase. Rezultate. Greutatea medie la naştere a fost mai mare după FET decât după ET (3316 g FET versus 3154 g ET). Rezultatul studiului a sugerat că protocolul FET a fost asociat cu rate semnificativ mai mici de greutate scăzută (16,8% FET versus 21,4% ET; p<0,05) şi cu rate mai mari de macrosomie (5,2% FET versus 0,8% ET; p<0,03), comparativ cu ET. Evaluarea a 216 nou-născuţi nu a arătat nicio diferenţă semnificativă statistic privind metodele de naştere (naştere prin cezariană sau naştere normală) sau sexul nou-născutului între cele două grupuri de transfer. Concluzii. Riscul de greutate mare la naştere în urma FET a fost mai mare decât cel după ET, dar riscul de greutate scăzută la naştere, dintre nou-născuţii din grupul FET, a fost semnificativ redus în comparaţie cu nou-născuţii din grupul ET.
Introduction
The application of in vitro fertilization (IVF) began 40 years ago(1). Daniel, the first IVF baby in Romania, was born 20 years later, in 1996. Romania was one of the tenderfoots to IVF in Europe because of its history of repressive reproductive policies(2). In vitro fertilization generally involves ovarian stimulation to produce a number of oocytes, followed by fertilization and fresh embryo transfer (ET), with surplus embryos frozen for transfer in subsequent cycles. Traditionally, IVF has involved the transfer of fresh embryos(3). Over the most recent decades, the use of frozen embryo transfer (FET) cycles has dramatically risen, including an 82.5% increase in use in the US between 2006 and 2012(4).
The primary reasons underlying the increasing trends for “freeze-all” are:
Vitrification technology has become the dominant method used for embryo cryopreservation, significantly increasing the embryo cryosurvival rate compared with slow-freezing(1). Vitrification, the alternative of traditional freezing for cryopreservation of embryos and oocytes, is regarded as a radical approach that totally eliminates ice formation and minimizes chilling injury, especially when the minimum volume – direct contact principle is applied(5). The first purposedly developed carrier tool, the Open Pulled Straw (OPS), has been used successfully in many species including humans. The special advantages of the OPS are the simple loading and expelling, the relative robustness, the consistent outcome and the possibility for sterile storage(6).
The freeze-all strategy is an approach that can be used to prevent ovarian hyperstimulation syndrome (OHSS).
Cryopreservation of embryos allows avoiding the detrimental effects of high-dose hormones used for ovarian stimulation on the endometrium. Additionally, the improved embryo-endometrium synchrony improves the chances of embryo implantation.
A wide variety of research has confirmed that FET may lead to more favourable perinatal and neonatal outcomes, reducing the risk of preterm birth and lower birth weight (LBW). However, FET has also been linked to large singleton infants, with increased rates of macrosomia and large-for-gestational-age(7,8,9).
The aim of the present study was to assess if the frozen-thawed embryo transfer (FET) results in better birth outcomes than fresh embryo transfer (ET).
In this study, we analyzed data from the IVF Centre of Târgu-Mureş for the following parameters: term, singleton infants, born between 2017 and 2019, following either ET or FET. We also examined the interaction between infant sex and frozen/thawed embryo transfer to determine if birth weight effects varied based on infant sex.
Materials and method
This retrospective study involved women undergoing in vitro fertilization between 2017 and 2019 using autologous eggs with singleton pregnancy delivered from the 37th week onward. Another inclusion criterion of the study was the normal BMI. The exclusion criteria included: cycles using donor oocytes or embryos, those in which the female patient was >40 years old at cycle start, cycles with missing data on infant sex and birth weight. Smokers were excluded as well.
Fetal macrosomia has been defined in several different ways, including birth weight greater than 4000-4500 g or greater than 90% for gestational age. In our study, birth weight was categorized as low (birth weight less than 2.5 kg) or high (birth weight greater than 4 kg). However, the risks associated with fetal macrosomia increase greatly when birth weight is more than 4,500 grams.
Our analysis restricted to cycles initiated from 2017 onwards to cover the time period when vitrification of embryos was increasingly adopted by our IVF laboratory as the standard practice. By applying these criteria, we found eventually 121 and 95 patients who underwent ET and FET, respectively.
All statistical analyses were performed using PAST 4 software, version 4.01. The differences were regarded significant with p values <0.05.
Results
We reviewed 216 women for the study period of two years. Maternal age was higher in the FET group than in the ET group (32.1 FET versus 31.9 ET).
The mean neonatal birth weight was higher for singletons born after FET than for singletons born after ET (3316 g FET versus 3154 g ET). The result of the trial suggested that the FET protocol was associated with lower rates of low birth weight (16.8% FET versus 21.4 % ET; p<0.05) and higher rates of high birth weight (5.2% FET versus 0.8% ET; p<0.03) than the ET protocol.
The assessment of 216 babies showed no statistically significant difference in the mode of delivery (42% C-section FET versus 43% C-section ET; p<0,375) and sex of live born between the two study groups (49.2% male infants FET versus 49.3% male infants ET; p<0.510).
Discussion
In this study we compared neonatal outcomes after FET with ET cycles, and our findings confirm that FET may lead to lower risks of low birth weight and to a higher neonatal birth weight than ET. A higher birth weight, lower rates of LBW and small-for-gestational-age (SGA), and higher rates of macrosomia and large-for-gestational-age (LGA) of singletons born after FET have been reported multiple times. For example, in a large Nordic cohort study, singletons born after FET had a lower risk of preterm birth (PTB), low birth weight, very PTB, and very PTB plus small-for-gestational-age than singletons born after ET(10). Additionally, more favorable neonatal outcomes via FET, including LBW and SGA rates, have been reported(11,12).
Only few studies have compared perinatal and maternal outcomes after vitrification and slow-freeze. According to some recent retrospective cohort studies, embryo vitrification was not associated with increased risks of low birth weight, small-for-gestational-age, large-for-gestational-age and macrosomia, as compared to slow-freezing(13,14).
Similar to Wennerholm et al. findings, in the current study we did not find any difference in sex ratio (male/female) between fresh cycle and FET group(15).
As far as we know, this is the first study of this type in Romania. The results of our study should be evaluated in the light of several limitations: this is a retrospective, small study from a single center, so a well-designed, prospective, randomized study with higher sample size is needed in the future to confirm our current findings. The information regarding pregnancy-related complications was unavailable. Maternal pregnancy-related complications, such as gestational hypertension and diabetes, can affect neonatal outcomes, such as neonatal weight, LBW, SGA, and large-for-gestational-age(16-18). Thus, whether poor neonatal outcomes – including LBW and high birth weight – are due to pregnancy-related complications or to differences in the type of ET technique used is unknown.
Conclusions
Our results indicate that the risk of high birth weight following frozen/thawed embryo transfer was greater than that following fresh embryo transfer, but the risk of low birth weight among frozen/thawed embryo transfer infants was significantly decreased in comparison with fresh embryo transfer infants.
The basic pathophysiology of increased risk of large-for-gestational-age and high birth weight in FET singletons remains uncertain. Several possible elements may additionally play a role – i.e., the parental characteristics, as well as the freezing-thawing procedures per se, which might induce epigenetic changes during early embryonic stages that alter the intrauterine growth capacity in FET offspring(19).
We should comprehensively inform patients about the advantages, disadvantages and the potential risks related to embryo cryopreservation(20,21).
Copyright Transfer Agreement
This manuscript has been approved by all authors.
This manuscript has not been submitted to another journal at the same time or has not already been accepted or published elsewhere.
The authors declare that they have no competing interests.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Wu J, Yang X, Huang J, Kuang Y, Wang Y. Fertility and Neonatal Outcomes of Freeze-All vs. Fresh Embryo Transfer in Women with Advanced Endometriosis. Frontiers in Endocrinology (Lausanne). 2019 Nov 8; 10:770, doi: 10.3389/fendo.2019.00770.
-
Michal R. Romanian IVF: a brief history through the ‘lens’ of labour, migration and global egg donation markets. Reproductive Biomedicine & Society Online. 2016 June; Volume 2, pages 79-87.
-
Zhang J, et al. Fresh versus frozen embryo transfer for full-term singleton birth: a retrospective cohort study. Journal of Ovarian Research. 2018; 11:59, https://doi.org/10.1186/s13048-018-0432-x.
-
Gliozheni O, Calhaz-Jorge C, De Geyter C, Kupka MS, de Mouzon J, Erb K, et al. Assisted reproductive technology in Europe, 2013: results generated from European registers by ESHRE. Hum Reprod. 2017; 32(10):1957–73.
-
Vajta G, Rienzi L, Ubaldi M. Open versus closed systems for vitrification of human oocytes and embryos. Reproductive BioMedicine Online. 2015; 30, 325–333.
-
Vajta G, et al. From a backup technology to a strategy – outlining approach: the success story of cryopreservation. Expert Review of Obstetrics & Gynecology. 2013; Volume 8, 181-190.
-
Shapiro BS, Daneshmand ST, Garner FC, Aguirre M, Hudson C, Thomas S. Evidence of impaired endometrial receptivity after ovarian stimulation for in vitro fertilization: a prospective randomized trial comparing fresh and frozenthawed embryo transfer in normal responders. Fertil Steril. 2011; 96:344–8, doi: 10.1016/j.fertnstert.2011.05.050.
-
Evans J, Hannan NJ, Edgell TA, Vollenhoven BJ, Lutjen PJ, Osianlis T, et al. Fresh versus frozen embryo transfer: backing clinical decisions with scientific and clinical evidence. Hum Reprod Update. 2014; 20:808–21, doi: 10.1093/humupd/dmu027.
-
Wu MY, Chung CH, Pan SP, Jou GC, Chen MJ, Chang CH, et al. Advantages of cumulative pregnancy outcomes in freeze-all strategy in high responders – A case-control matching analysis of a large cohort. J Formos Med Assoc. 2018; 117:676–84, doi: 10.1016/j.jfma.2018. 05.011.
-
Wennerholm UB, Henningsen AKA, Romundstad LB, Bergh C, Pinborg A, Skjaerven R, et al. Perinatal outcomes of children born after frozenthawed embryo transfer: a Nordic cohort study from the CoNARTaS group. Hum Reprod. 2013; 28:2545–53.
-
Wikland M, Hardarson T, Hillensjo T, Westin C, Westlander G, Wood M, et al. Obstetric outcomes after transfer of vitrified blastocysts. Hum Reprod. 2010; 25(7):1699–707.
-
Sazonova A, Kallen K, Thurin-Kjellberg A, Wennerholm UB, Bergh C. Obstetric outcome in singletons after in vitro fertilization with cryopreserved/thawed embryos. Hum Reprod. 2012; 27(5):1343–50.
-
Gu F, Li S, Zheng L. Perinatal outcomes of singletons following vitrification versus slow-freezing of embryos: a multicenter cohort study using propensity score analysis. Hum Reprod. 2019 Sep 29; 34(9):1788-1798.
-
Ginström Ernstad E, Spangmose AL, Opdahl S. Perinatal and maternal outcome after vitrification of blastocysts: a Nordic study in singletons from the CoNARTaS group. Hum Reprod. 2019 Nov 1; 34(11):2282-2289.
-
Vidal M, Vellve K, González-Comadran M, et al. Perinatal outcomes in children born after fresh or frozen embryo transfer: a Catalan cohort study based on 14,262 newborns. Fertility and Sterility. 2017 April; Vol. 107, No. 4, 940-947.
-
Yen TA, Yang HI, Hsieh WS, Chou HC, Chen CY, Tsou KI, et al. Preeclampsia and the risk of bronchopulmonary dysplasia in VLBW infants: a population-based study. PLoS One. 2013; 8(9): e75168.
-
Kissin DM, Jamieson DJ, Barfield WD. Monitoring health outcomes of assisted reproductive technology. N Engl J Med. 2014; 371(1):91–3.
-
Yee LM, Caughey AB, Cheng YW. Association between gestational weight gain and perinatal outcomes in women with chronic hypertension. Am J Obstet Gynecol. 2017; 217(3):348. e1-e9.
-
Orvieto R, Kirshenbaum M. Is Embryo Cryopreservation Causing Macrosomia – and What Else? Front Endocrinol. 2020; 11: 19.
-
Rienzi L, Gracia C, Maggiulli R, LaBarbera AR, Kaser DJ, Ubaldi FM, et al. Oocyte, embryo and blastocyst cryopreservation in ART: systematic review and meta-analysis comparing slow-freezing versus vitrification to produce evidence for the development of global guidance. Hum Reprod Update. 2017; 23(2):139–55.
-
Ishihara O, Araki R, Kuwahara A, Itakura A, Saito H, Adamson GD. Impact of frozen-thawed single-blastocyst transfer on maternal and neonatal outcome: an analysis of 277,042 single-embryo transfer cycles from 2008 to 2010 in Japan. Fertil Steril. 2014; 101:128–33.
Articole din ediţiile anterioare
Managementul neoplaziei cerebrale în timpul sarcinii
Cancerul cerebral în timpul sarcinii este o patologie rară, cu un prognostic rezervat, din cauza semnelor şi simptomelor care pot fi confundate c...