Noninvasive prenatal diagnosis, which has as the main purpose the detection of certain fetal genetic disorders, is possible due to the passage of fetal nucleated cells into maternal blood. It is important to know that there are studies that report important false-positive rates associated to these tests. We aimed to synthetize the main risk factors which may lead to the test failure through false-positive or false-negative results. Each of these factors should be taken into account when recommending noninvasive prenatal screening through fetal cell-free DNA analysis.
Factori implicaţi în eşecul testării prenatale neinvazive prin ADN extracelular fetal
Risk factors for fetal cell-free DNA testing failure – literature review
First published: 15 aprilie 2021
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.32.2.2021.4996
Abstract
Rezumat
Diagnosticul prenatal neinvaziv, care are drept scop principal detectarea anumitor anomalii genetice fetale, este posibil datorită trecerii celulelor nucleate fetale în sângele matern. Este important de ştiut că există studii care raportează o rată importantă de rezultate fals pozitive asociate acestor teste de screening. Astfel, ne-am propus să sintetizăm principalii factori de risc care pot duce la eşecul lor prin rate fals pozitive sau fals negative. Fiecare dintre aceşti factori trebuie luat în considerare la recomandarea screeningului prenatal neinvaziv, prin analiza ADN-ului fetal liber.
Introduction
Noninvasive prenatal diagnosis, which has as the main purpose the detection of certain fetal genetic disorders, is possible due to the passage of fetal nucleated cells into maternal blood(1). The detection of selected aneuploidies is performed by using next-generation sequencing of cell-free DNA in maternal circulation(2). The characteristic of cell-free DNA is represented by the fact that is derived from both maternal and fetal-placental unit and provides a very high specificity, with a detection rate of 99.5% for Down syndrome pregnancies, 97.7% for trisomy 18, and 96.1% for trisomy 13(3). The differences between noninvasive prenatal diagnosis, which is a screening test, and invasive methods like chorionic villus sampling or amniocentesis, which are diagnostic tests, relies on the presence of false-positive and false-negative results(4). All positive screening tests should be referred to an invasive prenatal diagnostic test. Noninvasive prenatal testing offers good prenatal results, but there are reports of an important failure rate that reaches about 10%(5). In the context of various values of failure rate reported, a consensus of 2% was decided(6). The false-positive rate and the detection rate are the main components of the screening performance; retesting, although, is not unusual in these cases. In high-risk pregnancies, invasive prenatal testing should be offered as an option. Obtaining an amount of fetal fraction DNA below an acceptable level is the most common reason for a test failure, this explaining about 50% of all failed results.
Materials and method
We analyzed the inconsistent results obtained after noninvasive prenatal testing, for a period of four years (2016-2019), in the Bucharest University Emergency Hospital, until the pandemic situation, as the COVID-19 impact on the health system limited the presentation for nonurgent health issues at multidisciplinary emergency hospitals.
Results
For a number of 503 cases, prenatal noninvasive testing was performed for the following reasons:
-
increased risk of aneuploidy determined by conventional first-trimester screening
-
individual maternal factors that determine an increased risk of aneuploidy
-
ultrasonographic abnormal findings, suggestive for the existence of chromosomal abnormalities.
For a number of 11 cases (2.18%), the obtained DNA fetal fraction was below 4%, and these patients were excluded from the further analysis.
Regarding the factors that influenced the value of the fetal fraction, we obtained an inversely proportional relationship between maternal weight and the average value of the DNA fetal fraction, with statistically significant value. Thus, the average of the fetal fraction logarithmically transformed, related to a maternal weight range of 50-55 kg, was 1.0172, finding a decrease of 0.0227 compared to an average increase in maternal weight of 5 kg. Pregnant women weighing less than 60 kg had a logarithmically transformed fetal fraction of approximately 1.0086, while those over 70 kg had a value of 0.9674, as presented in Figure 1.

Figure 2 includes a total percentage of 2.22% (11 cases) for the all four years, divided by the studied years, in which the increased risk of chromosomal abnormalities obtained after noninvasive prenatal testing was contradicted by the following invasive diagnostic tests. Thus, the limits of fetal DNA testing and, implicitly, its impossibility, at present, to replace the current diagnostic tests (amniocentesis/chorionic villus biopsy) are highlighted.
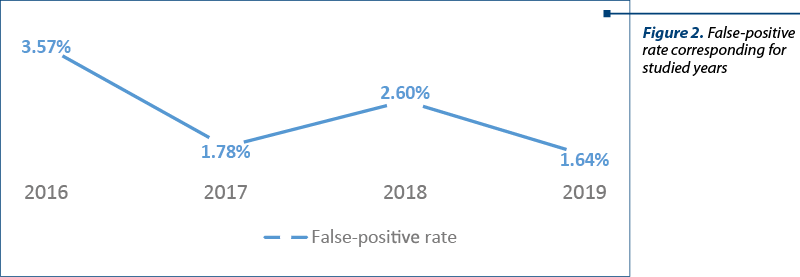
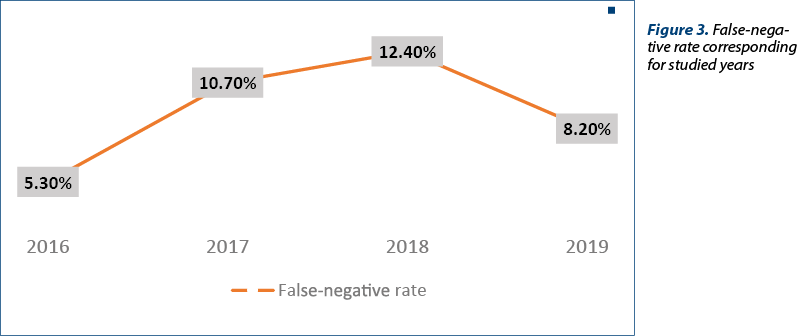
Figure 3 summarizes the number of patients who had a contradictory result of fetal DNA testing, later compared to that obtained from invasive prenatal testing, respectively the presence of a chromosomal abnormality in the context of a negative result of noninvasive prenatal testing. The increase in the percentage of contradictory results can be explained by the progress made by noninvasive prenatal testing in terms of the number of abnormalities detected, implicitly the sensitivity and specificity related to them. The results of our study, regarding the rate of detectability of chromosomal abnormalities in noninvasive prenatal testing, are similar to those reported in literature(3). Retrospectively analyzing the obtained results, it is worth mentioning that most of the false-negative results were associated to structural rearrangements, mosaicism or triploidies.
Discussion – specific factors related
to false-positive and false-negative results
Recent studies have shown that in pregnancies obtained through in vitro fertilization, compared with spontaneously obtained pregnancies, the median fetal fraction obtained was significantly lower. Test failure rate was higher and positive predictive value for trisomies 18 and 13 was lower in pregnancies obtained through in vitro fertilization(7). Other factors found to have implications in the amount of DNA fetal fraction are increased Body Mass Index (BMI) and low gestational age at testing. The performance of noninvasive prenatal testing in twin pregnancies was prospectively analyzed and the results showed that, in these cases, the median fetal fraction is significantly lower and, implicitly, the failure rate of the screening test is significantly higher(8). Also, the study demonstrated that maternal age is an important implicated factor, aside maternal Body Mass Index and crown-rump length of the fetus(9). It is to mention that the main factor involved in test failure in twin pregnancies was the conception through in vitro fertilization(10).
Important long fragments of DNA homozygosity, characterized by identical fragments of gene sequencing from maternal and paternal origin, are also a reason for test failure. In cases of parental consanguinity or uniparental disomy, the results obtained will be in most cases borderline, which should be considered a screen positive test and follow-up actions should be initiated(11).
In multiple cases, the abnormal results are centered only in one assay, respectively the interpretation for sex chromosome related syndromes or microdeletion syndromes, and rarely includes total assay failure(12).
In objective terms, if we consider that there is a 10% false-positive rate for cell-free DNA screening, meaning that 10% of the fetuses screened as positive will be unaffected, this percentage looks enormous. However, screening for trisomy 21 by analysis with cell-free DNA in maternal blood provides superiority in results regarding the detection rate and false-positive rate compared to all other traditional methods of screening. It is to be considered also the fact that the performance of screening for trisomy 13, 18 and sex chromosome aneuploidies is importantly lower compared to the results for trisomy 21(12). In Table 1, the main reasons for a false-positive result of cell-free DNA testing are centralized.
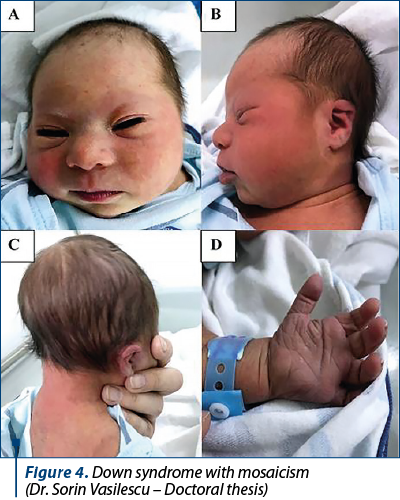
In case of placental mosaicism, false-positive cell-free DNA test result may occur in 1-2% of pregnancies, especially for monosomy X and trisomy 13(13). The background of this failure rate consists in the fact that the main source of fetal cell-free DNA in maternal blood originates from the syncytiotrophoblast, therefore the test results will be specific for the placenta mainly, that cannot be concordant with the fetal tissue(13).

In twin pregnancies, if the demise of one embryo occurs very early, this complication can pass unnoticed. The false-positive rate in such cases relies on the fact that the placenta of the vanished twin, which has a high possibility to be aneuploidy, continues to shed DNA in maternal circulation a significant period after demise(9). An important study that included 30,797 singleton pregnancies who underwent noninvasive prenatal testing for whole-chromosome aneuploidies showed that the fewer false-positive rate can be obtained by identifying additional fetal haplotypes(7).
Generally valid hypothesis of most noninvasive prenatal tests is that the mother has a normal karyotype. However, it is well known that advanced age in the majority of women consists in a small amount of monoploid mosaic that will be attributed to the fetus, leading to a false-positive cell-free result for sex chromosomes aneuploidies. In such cases, the diagnosis can be clarified by karyotyping peripheral blood lymphocytes.
Carlson et al. published in 2018 an article where they implied the possibility of detection of various maternal condition starting from the false-positive results of cell-free DNA tests. Thus, if more than one aneuploidy is detected that is discordant with the fetal karyotype, than the risk of maternal malignancy is significantly elevated(14).
If the size of maternal DNA duplication was larger than average and it occurs on a chromosome of interest, cell-free DNA sequencing may give a false-positive result(15).
Another important issue are the false-negative results, a fact that can have even higher disturbance on parent’s decision. The main reason why these results can appear is the placental mosaicism when the main fetal fraction is represented by placental cells which may not clinically correspond with the fetal tissue. If the amount of fetal fraction is borderline (3-5%), an insufficient number will be sequenced and the result will be false negative(15).
Cell-free DNA screening is commonly used as a secondary screening test for trisomies 21, 18 and 13, respectively, offered to a population that already has a high or intermediate positive risk attributed from the first-trimester prenatal screening, being an alternative that reduces significantly the number of unnecessary invasive procedures.
Conclusions
Several key points are recommended to be followed when offering cell-free DNA as a screening test. It is important to offer a genetic counseling to the parents before testing. This counseling must contain the main information about test principles, the meaning of a screening test and the fact that is optional, information about the pathologies that can be found and about the results that can be obtained. Also, the fact that this test has limitations and the possibility of the need of other diagnostic tests before deciding to end the pregnancy should be also clarified.
In Romania, the cost of cell-free DNA testing is not covered by insurance policy, but there are national and local programs that can help with financial support.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Taylor-Phillips S, Freeman K, Geppert J, et al. Accuracy of non-invasive prenatal testing using cell-free DNA for detection of Down, Edwards and Patau syndromes: a systematic review and meta-analysis. BMJ Open. 2016;6(1):e010002.
-
Gil MM, Accurti V, Santacruz B, et al. Analysis of cell-free DNA in maternal blood in screening for aneuploidies: updated meta-analysis. Ultrasound Obstet Gynecol. 2017;50(3):302-14.
-
Lee TJ, Rolnik DL, Menezes MA, et al. Cell-free fetal DNA testing in singleton IVF conceptions. Hum Reprod. 2018;33(4):572-8.
-
Munteanu O, et al. The etiopathogenic and morphological spectrum of anencephaly: a comprehensive review of literature. Rom J Morphol Embryol. 2020;61(2):335-43.
-
Voicu D, Munteanu O, Sajin M, Dumitru A, Darabont R, et al. Pregnancy outcomes in a patient with Fontan circulation for single ventricle congenital heart disease and aberrant praevia placental lobe. Rom J Military Med. 2019;3:110-14.
-
Păuleţ F, Ţurcan N, Gherghiceanu F, Bohîlţea RE, Nemescu D, Cîrstoiu MM. Prognosis of autoimmune thyroid disease associated with hereditary thrombophilia during pregnancy. Exp and Ther Med. 2020;20(3):2429-33.
-
Sarno L, Revello R, Hanson E, et al. Prospective first-trimester screening for trisomies by cell-free DNA testing of maternal blood in twin pregnancy. Ultrasound Obstet Gynecol. 2016;47(6):705-11.
-
Bohîlţea RE, Zugravu CA, Neacşu A, Navolan D, Berceanu C, Nemescu D, Bodean O, Ţurcan N, Baros Al, Cîrstoiu MM. The prevalence of Vitamin D deficiency and its obstetrical effects. A prospective study on Romanian patients. Rev Chim. 2019;70(4):1228-33.
-
Cîrstoiu MM, Filipoiu FM, Brătilă E, Berceanu C, Cîrstoiu FC, Budu VA, Bulescu IA, Munteanu O. Morphological study of cephalothoracopagus deradelphus type conjoined twins. A case report. Rom J Morphol Embryol. 2016;57(1):249-52.
-
Gil MM, Quezada MS, Revello R, et al. Analysis of cell-free DNA in maternal blood in screening for fetal aneuploidies: updated meta-analysis. Ultrasound Obstet Gynecol. 2015;45(3):249-66.
-
Malvestiti F, Agrati C, Grimi B, et al. Interpreting mosaicism in chorionic villi: results of a monocentric series of 1001 mosaics in chorionic villi with follow-up amniocentesis. Prenat Diagn. 2015;35(11):1117-27.
-
Curnow KJ, Wilkins-Haug L, Ryan A, et al. Detection of triploid, molar, and vanishing twin pregnancies by a single-nucleotide polymorphism-based noninvasive prenatal test. Am J Obstet Gynecol. 2015;212(1):79.e1-9
-
Wang Y, Chen Y, Tian F, et al. Maternal mosaicism is a significant contributor to discordant sex chromosomal aneuploidies associated with noninvasive prenatal testing. Clin Chem. 2014;60(1):251-9.
-
Carlson LM, Hardisty E, Coombs CC, Vora NL. Maternal malignancy evaluation after discordant cell-free DNA results. Obstet Gynecol. 2018;131(3):464-8.
-
Bohîlţea R, Ţurcan N, Cavinder CM, et al. Risk factors, predictive markers and prevention strategies for intrauterine fetal death. An integrative review. J Mind Med Sci. 2020;7(1):Art 10.
Articole din ediţiile anterioare
Testarea genetică prenatală neinvazivă prin AND liber fetal - metodă de screening pentru sarcinile cu risc înalt de malformaţii fetale
Domeniul screeningului şi al diagnosticului genetic prenatal neinvaziv pare a fi revoluţionat de introducerea testării ADN-ului fetal liber din sân...
Uterine carcinosarcoma – histological and immunohistochemical features
Carcinosarcoma is a biphasic neoplasm with two distinct malignant tissue types, carcinomatous and sarcomatous. The epithelial component can develop...
Rolul examenului ecografic în evaluarea cauzelor ovariene de infertilitate
Infertilitatea este în general definită ca inabilitatea de a concepe după un an de relaţii sexuale neprotejate, dar în cuplurile cu vârsta de peste...