From an immunological point of view, pregnancy is a physiological condition treated by the body as a semiallograft, the product of conception holding 50% of genetical information from the partner. Hormonal and biochemical changes specific to pregnancy lead to decreased immunity in order to tolerate fetal development. This immune tolerance causes an improvement in most autoimmune diseases during pregnancy. However, there are some exceptions to this rule, therefore in this article we present the case of a pregnant woman who developed an exacerbation of the clinical manifestations of pemphigus during pregnancy. The informed consent was obtained from the patient for the publication of this case. We chose to present this case due to the rare incidence of pregnancy-associated pemphigus.
Pemfigus asociat sarcinii – prezentare de caz
Pregnancy-associated pemphigus – case report
First published: 31 mai 2022
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.36.2.2022.6549
Abstract
Rezumat
Din punct de vedere imunologic, sarcina reprezintă o condiţie fiziologică tratată de către organism ca o semiallogrefă, produsul de concepţie deţinând 50% din informaţia genetică a partenerului. Modificările hormonale şi biochimice specifice perioadei de graviditate duc la scăderea imunităţii în scopul de a tolera dezvoltarea fetală. Această toleranţă imunologică determină o ameliorare pe perioada sarcinii a majorităţii bolilor autoimune. Există însă şi excepţii de la această regulă, astfel că, în acest articol, vom prezenta cazul unei parturiente care a dezvoltat o exacerbare a manifestărilor clinice de pemfigus pe durata sarcinii. Consimţământul informat a fost obţinut de la pacientă pentru publicarea acestui caz. Am ales să prezentăm acest caz datorită incidenţei reduse a pemfigusului asociat sarcinii.
Introduction
Pemphigus vulgaris (PV) is a rare and serious (potentially life-threatening) condition that causes painful blisters on the skin and mucous membranes of the mouth, nose, throat and genitals. The blisters are fragile and can open easily, leaving bare areas of the skin, which are very painful and create a risk of infection. There is currently no cure for pemphigus vulgaris, but the treatment can help maintain the symptoms under control. The condition can affect people of all ages, including children, but most cases develop in older adults between the ages of 50 and 60. It is not contagious and cannot be passed from one person to another.
Case presentation
A 42-year-old woman, C.D.D., with a 36-week pregnancy, presented to the emergency room for generalized rash accompanied by severe pruritus, a rash that had started about two months before, with worsening of the symptoms in the last 24 hours.
The patient stated that she had been previously diagnosed with polycystic ovaries syndrome, without following any treatment in this regard. The patient also stated that she was a non-smoker because she was pregnant, and that she had no known food or drug allergies.
From the anamnesis, we found out that the symptoms had started insidiously, about two months before. The patient said that the appearance of generalized skin lesions started in the upper limbs with gradual dissemination in the lower limbs, chest and abdomen – intensely itchy. She was hospitalized with suspected cholestasis of pregnancy, receiving treatment with deoxycholic bear acid 250 mg, 2 tb/day, without any improvement in symptoms; later, the patient was referred to the internal medicine and dermatology service, raising the suspicion of gestational pemphigus and underwent treatment at home with corticosteroids-like prednisone 5 mg, 6 tb/day, omeprazole 40 mg and cetirizinum 10 g orally, with a slight relief of symptoms.
The pregnancy was partially dispensed, and the patient did not perform the morphology of the first and second trimesters, the first ultrasound being performed at 21 weeks of gestation. During hospitalization, we noticed increased values of bile acids (7.4 umol/dL), decreasing values after the administration of ursodeoxycholic acid (2.5 umol/L), fibrinogen (490 mg/dL) and increased ESR (39 mm/h). Immunology: negative for antimitochondrial (AMA) and antinuclear (ANA), anticytoplasmatic (ANCA) and rheumatoid factor antibodies. Also, negative serology for liver viruses B and C, herpes, HIV, syphilis, Listeria and TORCH complex.
An incompatibility of Rh was identified, with absent antibodies dosed at 30 weeks of gestation.
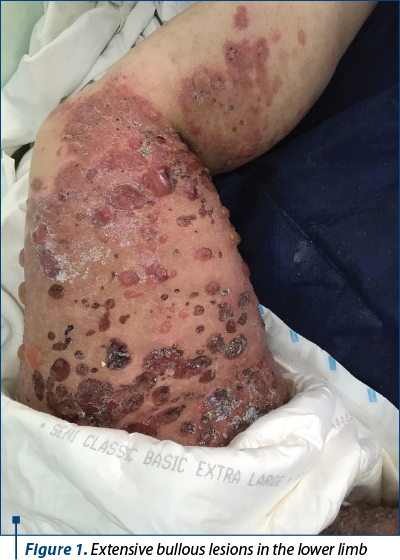
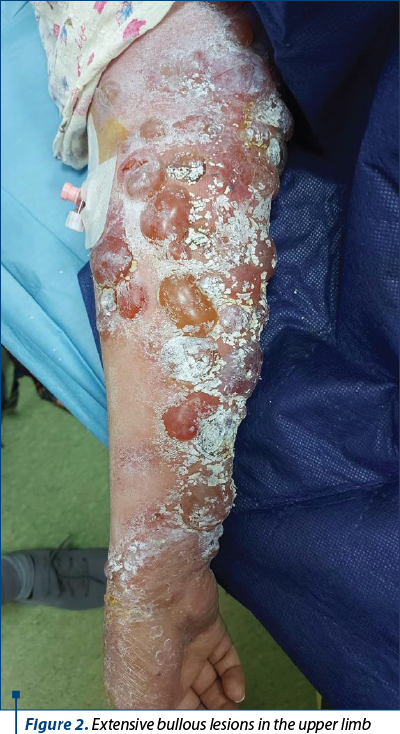
We note that the patient refused to perform the skin biopsy, after the recommendation of the dermatologist.
General clinical examination
Stable hemodynamically and respiratory patient, afebrile, 120/70 mmHg, pulse rate 74 bpm, rhythmic, temporally-spatially oriented, cooperating, intestinal transit present, spontaneous urination, weight about 90 kg, with a weight gain during pregnancy for about 15 kg.
At the level of the upper and lower limbs, chest and abdomen, there were extensive, generalized lesions that respected the face and erythematous plaques arranged on the abdomen and flanks. She had an increased abdomen, long longitudinal axis and hyperpigmentation of the white abdominal line.
Through Leopold’s maneuvers, we palpated a hard, non-depressible area, with a regular contour, suggestive for the fetal skull at the lower pole; a depressible formation was palpated at the upper pole, suggestive for the fetal pelvis, a hard, concave plane with a surface was palpated in the right uterine flank, smooth, suggestive for the fetal back, a presentation confirmed later through abdominal ultrasound. Auscultation: rhythmic FCB = 137 bpm, highlighted on the right spine-umbilical line.
The valve examination showed a cervix with OCE without lesions, a normal macroscopic appearance, minimal leukorrhea. Vaginal touch: shortened cervix, closed and posterior.
The transabdominal obstetric ultrasound performed with difficulty suggested a single pregnancy, evolving, a live fetus, in cranial presentation, with rhythmic FCB of 133 bpm, AFM present, a normally inserted placenta, posterior and fundic, grade II of maturation, with no signs of detachment present at the time of examination, amniotic fluid in normal quantity, AFI 16.71, Doppler velocimetry in normal parameters, fetal biometrics corresponding to gestational age, and the fetal weight was estimated at approximately 2,500 g.
The hem leukogram showed leukocytosis with neutrophilia (15.7) and discrete eosinophilia (1.94), mild anemia (10.4) and thrombocytosis (419). Biochemistry: hypercholesterolemia (302 mg/dL), elevated triglycerides levels (329 mg/dL), elevated GGT (59 mg/dL), a normal urine test, and the other tests without pathological changes.
CTG performed at hospitalization: reactive, with variability, we noted the presence of accelerations, a basic rhythm at 120-140 bpm, active fetal movements, absence of systematized uterine contraction, normal uterine tonus.
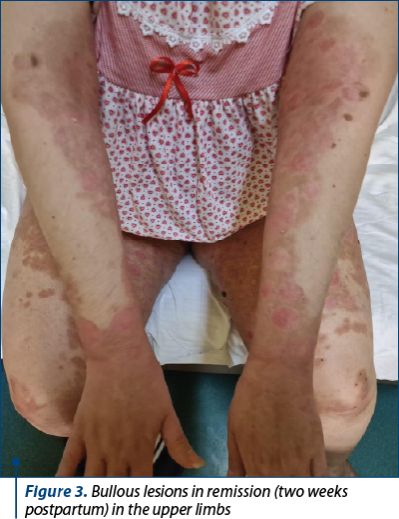
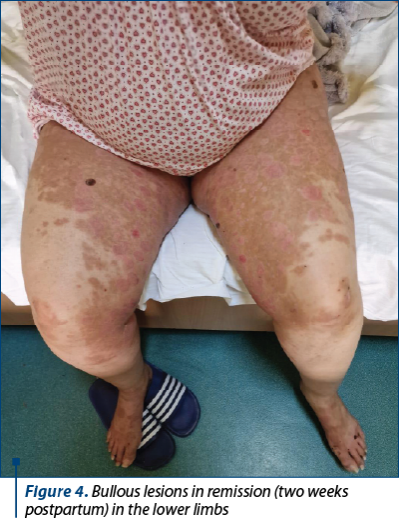
A dermatological examination was requested in the emergency room, with the establishment of the presumptive diagnosis of gestational associated pemphigus – severe generalized form.
Positive diagnosis: IIG, IP pregnancy, at 34 weeks, live fetus, in cranial presentation, with intact amniotic membranes, gestational associated pemphigus.
The suspicion of gestational pemphigus was supported by the nature of the skin lesions associated with pruritus, with an insidious onset in the third trimester of pregnancy, the patient refusing the skin biopsy.
Differential diagnosis
Before blistering, the lesions may be similar to those of PPUPP (pregnancy pruritic urticarial papules and plaques), pustular pregnancy psoriasis, herpetiform dermatitis, erythema multiforme, linear Ig A bullous dermatosis, urticaria, allergic contact dermatitis, or Stevens-Johnson syndrome.
Referring to gestational pemphigus, the gold standard in diagnosis this condition remains the direct immunofluorescence of the tissue fragment, skin biopsy and serum antibody dosing.
Discussion
Gestational pemphigus is an autoimmune, bullous, rare condition characterized by the appearance of pruritic papules and urticarial plaques, usually followed by the appearance of blisters in 1-2 weeks, mostly periumbilical, that respect the face, mucous membranes and scalp, being the result of a primary reaction between the maternal IgG antibodies directed against the XVII collagen from the structure of the basal membrane of the skin and amniotic epithelium.
There is an association between the described pathology and the restriction of fetal intrauterine growth and/or premature birth due to placental insufficiency by deposition of immune complexes in the amniotic basement membrane, low fetal weight at birth, and ulcer-necrotic enterocolitis.
In this case, considering the severity of the lesions induced by the current pathology associated and exacerbated by pregnancy, we definitely decided to hospitalize the patient for obstetric and dermatological evaluation. Echocardiographic monitoring twice a day – fetal ultrasound with evaluation of fetal growth, placental condition, AF volume (AFI), daily Doppler velocimetry of AO and ACM. We counseled the pregnant woman regarding the latency period and the psychological damage induced by pruritus and generalized lesions.
Corticosteroid therapy is initiated with dexamethasone 8 mg i.m./i.v. at 12 hours and prophylactic antibiotic therapy with amoxicillin 875 mg/clavulanic acid 125 mg, 1 tb/12 hours, for five days. Local skin application with Dermobacter® solution, twice a day, and repeating the laboratory tests, respectively HLG, choagulogram, urea, creatinine, D-dimers, transaminases, LDH and fibrinogen, at 48 hours.
At 36 hours after hospitalization, the patient spontaneously presented a watery leukorrhea, in abundant quantity, and after the local examination with valves, spontaneously ruptured membranes were noticed, the vaginal touch highlighting an incomplete erased cervix, with wide index finger.
Given the severity of the case and the location of the skin lesions, but reaching to the age of fetal viability (more than 34 weeks of gestation), as well as the complete scheme of dexamethasone treatment for maternal and fetal interest, it was decided to initiate the drug triggering of the labor and complete the birth naturally.
If CTG monitoring shows any signs of fetal distress accompanied by fetal hemodynamic damage (reversal of velocimetric flow or increased index on the umbilical artery), caesarean section is recommended – an extremely difficult approach due to the extensive injuries throughout the abdomen.
The maternal prognosis is generally good; postpartum, the skin lesions heal slowly, without leaving scars in most of the patients, in about three months after birth. In this case, the parturient was transferred to the dermatology department with a significant reduction in skin lesions at 10 days postpartum.
Regarding fetal prognosis, in about 10% of newborns, IgG-type antibodies can be passively transferred from the mother, causing similar skin lesions (which happened in this case, as well), these lesions requiring only local care and disappearing spontaneously in a few weeks after birth.
Conclusions
An elderly primiparous patient, with partially followed pregnancy, without known autoimmune components outside the discussion of the etiology of the micropolycystic ovary, developed gestational pemphigus with severe, generalized bullous lesions, without significant IUGR fetal impairment (2120 g at 34 weeks of gestation), but with the determination of a premature birth in maternal and fetal interest. We noticed the passive transfer of maternal antibodies to the fetus, with the appearance of fetal skin lesions, but also the regression of maternal skin lesions after birth.
We also highlight the triggered labor by its natural evolution, but also the need for psychological counseling of the patient who has been affected by injuries that have greatly altered her well-being.
Conflict of interests: The author declares no conflict of interests.
Bibliografie
-
Adams KM, Yan Z, Stevens AM, Nelson JL. The hypothesis of changing the maternal “self”: a mechanism of maternal tolerance of the foetus. Placenta. 2007;28:378–82.
-
Anolik JH. B cell biology and dysfunction in SLE. Bull NYU Hosp Jt Dis. 2007;65(3):182–6.
-
Bonilla WV, et al. Microchimerism maintains the deletion of the CD8+ T cell repertoire specific to the donor cell. J Clin Invest. 2006;116(1):156–62.
-
Branch DW, Porter TF. Autoimmune disease in pregnancy. In: PJ, James DK, Weiner CP, Gonik B (editors). High-risk task management options. 2nd ed., 1999:853–4.
-
Jacob CO, et al. Major hereditary histocompatibility class II-associated complex differences in the production of tumour necrosis factor alpha: relevance for the genetic predisposition to systemic lupus erythematosus. Proc Natl Acad Sci USA. 1990;87(3):1233–7.
-
Jiang SP, Vacchio MS. Several mechanisms of tolerance of peripheral T cells in fetal “allograft”. J Immunol. 1998;160:3086–90.
-
Nelson JL, Ostensen M. Pregnancy and rheumatoid arthritis. Rheum Dis Clin Nord Am. 1997;23(1):195–212.
-
Nelson JL, et al. Fertility before the onset of the disease in women with rheumatoid arthritis. Rheum Arthritis. 1993;36(1):7–14.
-
Nelson JL, et al. Maternal-foetal disparity in class II HLA alloantigen’s and pregnancy-induced amelioration of rheumatoid arthritis. N Engl J Med. 1993;329(7):466–71.
-
Nelson JL, et al. Microchimerism and HLA-compatible relationships of pregnancy in scleroderma. Lancet. 1998;351(9102):559–62.
Articole din ediţiile anterioare
Provocarea trombofiliei: profilul pacientelor cu trombofilii compuse rare
Trombofilia este o patologie multigenică. Mutaţia factorului V Leiden şi mutaţia genei protrombinei G20210A sunt cele mai frecvente trombofilii ere...
Tumoră spinală cervicală asociată sarcinii
Tumorile spinale sunt rar întâlnite în sarcină, dar atunci când sunt extinse, pot cauza probleme grave prin efectele neurologice rapid progresiv...
Diabetul gestaţional – o provocare obstetricală, neonatală şi postnatală
In recent decades, the demographics of pregnant women have changed, with an increase of women giving birth at an older age and which, associated wi...
Managementul unui paciente cu hemofilie, formă dobândită gestaţional – prezentare de caz
Hemofilia A dobândită este o patologie hemoragică rară şi severă în care organismul produce anticorpi specifici împotriva factorului VIII al coagul...