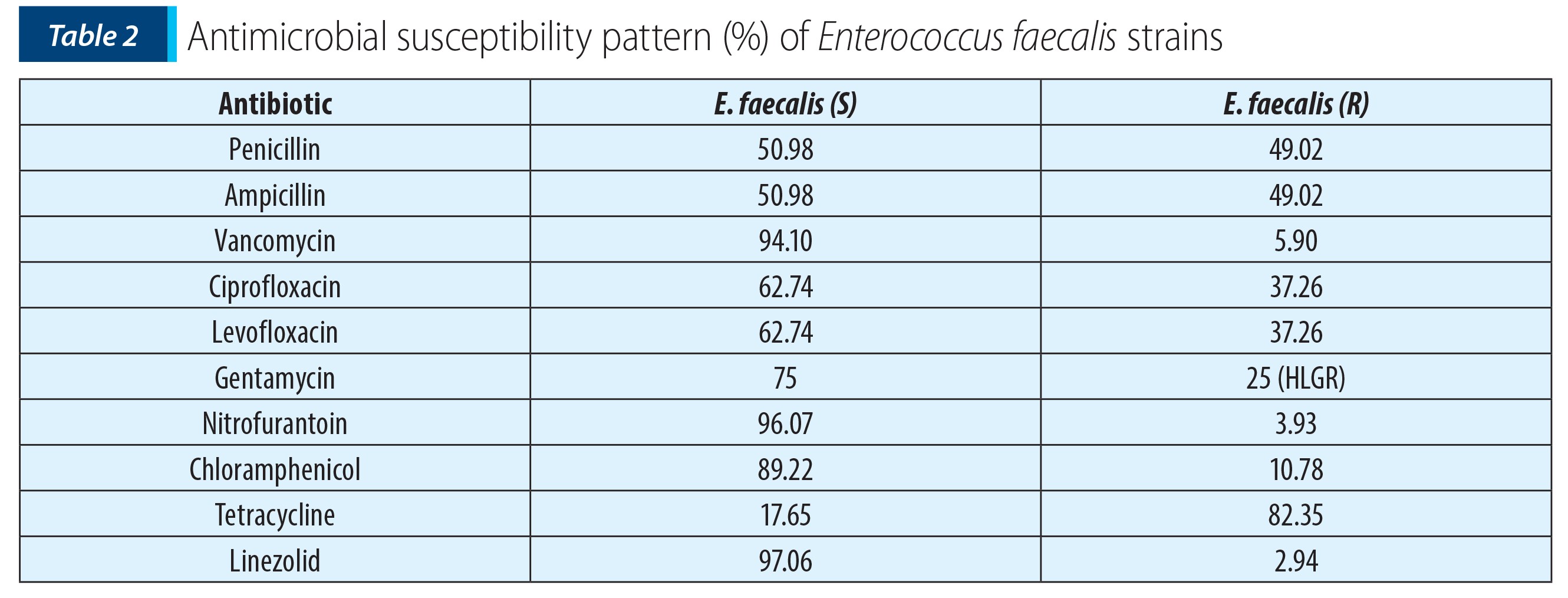
Enterococci are one of the important causes of urinary tract infections and are inherently resistant to many antimicrobials, including penicillin, clindamycin, trimethoprim-sulfamethoxazole and low levels of aminoglycosides. Moreover, they are poorly responsive to cephalosporins in vivo. The study was carried out in “Cantacuzino” National Research Institute on samples received from community. In order to detect resistance in Enterococcus species, a total of 102 Enterococcus species samples were included in the study. Although Enterococcus faecalis (98.03%) was one of the most common species associated with urinary tract infections, we also identified Enterococcus faecium (1.97%), showing the necessity of species level identification in laboratory settings. There were also isolated enterococci from mixture of two or three bacteria. E. faecalis was sensitive to vancomycin (94.10%), ampicillin (50.98%), penicillin (50.98%), linezolid (97.06%), nitrofurantoin (96.07%), levofloxacin (62.74%), ciprofloxacin (62.74%), chloramphenicol (89.22%) and tetracycline (17.65%), and resistant to gentamycin – high level resistance (25%).
Infecţii enterococice ale tractului urinar, analizate în Institutul Naţional de Cercetare „Cantacuzino”
Enterococcal urinary tract infections analyzed at “Cantacuzino” National Research Institute
First published: 06 noiembrie 2017
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Inf.51.3.2017.1198
Abstract
Rezumat
Enterococii constituie una dintre importantele cauze ale infectiilor tractului urinar si sunt rezistenti la multe antibiotice, incluzand penicilina, clindamicina, trimethoprim-sulfamethoxazolul si la niveluri scazute de aminoglicozide. In plus, raspund slab la cefalosporinele administrate in vivo.
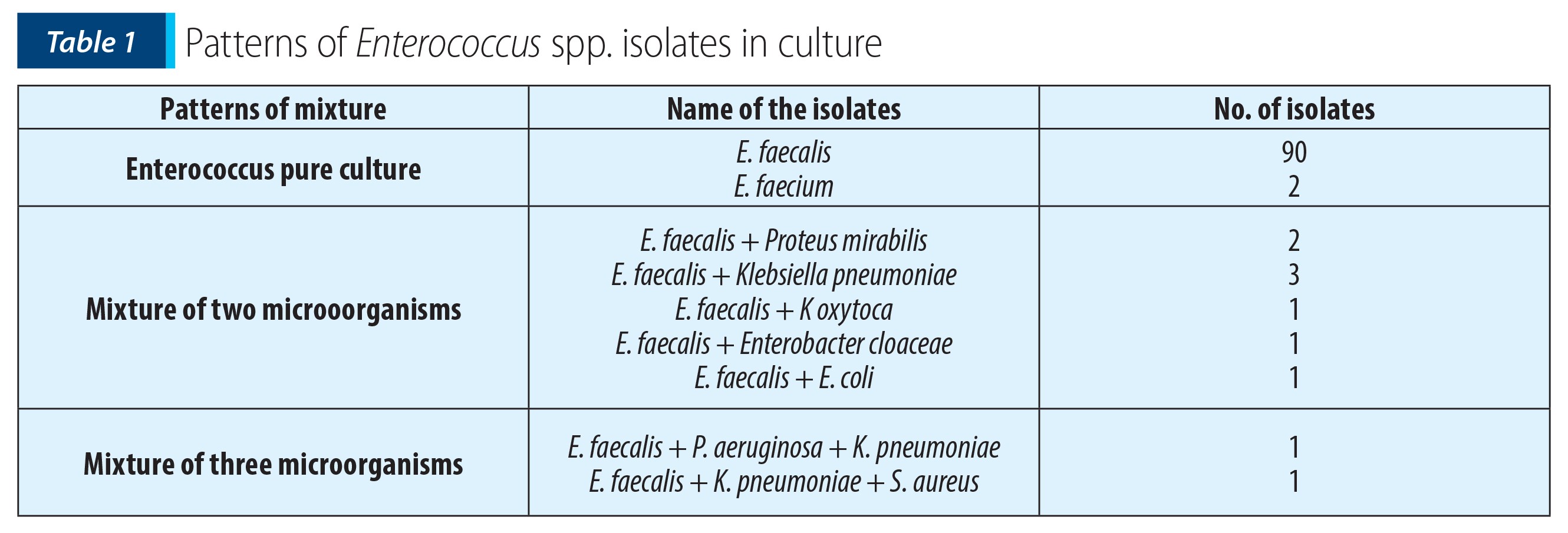
Studiul a fost executat la Institutul National Cercetare „Cantacuzino” testând prelevate primite din comunitate (ambulator), pentru a detecta rezistenta la speciile de Enterococcus. O suta doua prelevate din genul Enterococcus au fost incluse in studiu. Cu toate că Enterococcus faecalis (98,03%) a constituit una dintre speciile asociate in mod obisnuit cu infectiile tractului urinar, am identificat şi specia Enterococcus faecium (1,97%), ceea ce demonstrează necesitatea identificarii la nivel de specie in cadrul laboratoarelor. De menţionat că au fost izolati enterococi şi din culturi polimicrobiene (2 sau 3 bacterii).
E. faecalis a prezentat sensibilitate faţă de vancomicină (94,10%), ampicilină (50.98%), penicilină (50,98%), linezolid (97,06%), nitrofurantoin (96,07%), levofloxacin (62,74%), ciprofloxacin (62,74%), cloramfenicol (89,22%), tetraciclină (17,65%) şi rezistenţă de nivel înalt faţă de gentamicină (25%).
Introduction
Enterococci are part of the normal faecal flora in humans, but they also colonize the oral cavity, the genitourinary tract and the skin, particularly in the perianal area(1). The main sites of colonization in the hospitalized patients are soft tissue wounds and ulcers and the gastrointestinal tract(2). They were traditionally regarded as low grade pathogens, but have emerged as second leading cause of nosocomial infections and third most common cause of bacteraemia(3).Urinary tract infection (UTI) is the most common cause of nosocomial infection among hospitalised patients(4). In spite of the fact that enterococci are one of the most frequent aetiology identified, there are still a lot of characteristics that need to be established(5). Intra-abdominal and intra-pelvic abscesses or post surgery wound infections(5) and blood stream infections(3) are also commonly caused by Enterococcus next to UTIs.
Enterococci are classified in five goups, of which only a small part are responsible for producing different pathologies(6), the most frequent identified species being Enterococcus faecalis and Enterococcus faecium(7).
Enterococci are considered important especially for their ability of acquiring antibiotic resistance(7). High Level Aminoglycoside Resistant Enterococci (HLAR) were first reported in France in 1979 and since then they have been isolated from all the continents(8). Enterococcal infections are frequently identified in patients with bacteremia, surgical site infections or others, requiring combinations of antibiotics such as aminoglycosides with beta lactam(9). Since enterococci that show a HLAR phenotype are not susceptible to aminoglycosides, this antibiotic could not be used for the combination therapy(10).
Resistance to β lactam was first reported in early 1980s. Therefore it necessitates the higher dosage of β lactam and related drugs(1). So, detection of β lactam resistant enterococci should be reported. Resistance to tetracycline, erythromycin and chloramphenicol is well known nowadays. This study was aimed to determine the prevalence of multidrug-resistant uropathogenic Enterococcus.
Materials and method
The study was conducted in “Cantacuzino” National Research Institute, from May 2015 to September 2017. A total of 102 enterococcal isolates were included in the study.
Methodology
UTI was defined as the presence of >105 colony forming units/ml in the culture of an appropriate collected urine specimen. Standard microbiological techniques were used.The identification of Enterococcus spp. was done using the following parameters: (i) colony morphology on blood agar(11), (ii) Gram staining, (iii) catalase, (iv) bile esculin hydrolysis, (v) salt tolerance (growth in 6.5 % NaCl brain heart infusion). Then speciation was performed by sugar fermentation, reduction of tellurite in tellurite blood agar plate and analysis on API Strep kit. All tests were carried out and interpreted as described by Facklam and Collins(12).
Resistance profile
Antibiotic susceptibility pattern: antimicrobial susceptibility test was performed by Kirby-Bauer disc diffusion method. The peptone water culture standardized to 0.5 McFarland opacity was used for surface seeding on Mueller Hinton agar. After plates were dried, antibiotic discs were placed over the medium and incubated at 37°C for 24 hours. Then the results were recorded according to National Committee for Clinical Laboratory Standards(13) for the following antibiotic discs: vancomycin (30 μg), ampicillin (10 μg), penicillin (10 units), high level gentamycin (120 μg), ciprofloxacin (5 μg), levofloxacin (5 μg ), chloramphenicol (30 μg), tetracycline (30 μg), nitrofurantoin (300 μg), linezolid (30 μg). E. faecalis ATCC 29212, Staphylococcus aureus ATCC 25923 and E. faecalis ATCC 51299 were used as control strains(13).In order to detect High Level Gentamicin resistance (HLGR), minimum inhibitory concentration (MIC) of gentamicin was detected for the 27 isolates which showed resistance to high level gentamycin discs (120 μg). The bacterial inoculums of 1.5 x 108 CFU/ml was prepared with brain heart infusion broth, stock solution of gentamycin 5120 μg/ml was prepared with distilled water, to which 9 ml of overnight culture of brain heart infusion broth was added. The method of two fold dilutions up to 1 μg/ml was used. One control culture broth without antibiotic was also kept. After overnight incubation at 37°C with ambient air, the tubes were examined. The point at which there was no visible growth was taken as the minimum inhibitory concentration of gentamycin(14).
Results and discussions
It is important to know the changing trends of the Enterococcus infections and their antimicrobial susceptibility pattern. In our study, 102 enterococcal strains were recovered from urine specimen.Out of 102 isolates, 44 were from male and 58 were from female patients. About 48.03% of isolates were recovered from patients below 50 years of age. In a study from 2011, Kafayat et al. found that enterococcal infections in the age group of 20-29 years old represented the largest proportion (42.4%), followed by age group of 30-39 years old (23.7%)(15). This was supported by a survey done by Centers for Disease Control and Prevention (CDC) on nosocomial infections, in which Enterococcus accounted for 13.9% infections, being next to Escherichia coli as a causative agent of hospital acquired urinary tract infections(16).
Although the recent studies stated that there is an increase in the isolation of E. faecium and other enterococcal species(17), in our study E. faecalis (98.03%) constituted the major isolate, followed by E. faecium (1.97%). Similar findings were revealed by Facklam et al(18). Enterococcus spp. were isolated in pure culture (90.19%), recovered with other organisms as mixture of two organisms (7.84%) or three organisms (1.96%). Enterococcus faecalis was associated with other enterobacterial species (Table 1).
E. faecalis isolates were susceptible to vancomycin (94.10%), ciprofloxacin (62.74%), levofloxacin (62.74%), ampicillin (50.98%), penicillin (50.98%), tetracycline (17.65%), chloramphenicol (89.22%), linezolid (97.06%), nitrofurantoin (96.07%) and revealed HLR to gentamycin (25%) (Table 2).
Two strains (E. faecium) showed resistance to ciprofloxacin, one of the most common antibiotic used to treat urinary tract infection, as well as to gentamycin, and were susceptible to vancomycin and linezolid. On the other hand, 49.02% of the E. faecalis isolates showed resistance to ampicillin. HLGR (120 μg/ml) by disk diffusion method was observed in 25% isolates. HLGR was referred as MIC>500 μg/l(19), and was seen in 26.47% of isolates. Sreeja S et al. (2012) found 45% resistance to ampicillin, 50% to ciprofloxacin and 47% to high level gentamicin among Enterococcus isolates(20). A study by Kapoor et al. (2005) found 66% HLAR(21), and Vittal P. Prakash et al. (2005) found 43.5% HLGR(22).
Although E. faecalis (98.03%) was the most common species identified in clinical specimens, there was a deviation towards E. faecium isolation (1.97%). Further, there is an increased isolation of multidrug-resistant Enterococcus with HLGR (26.47%). Thus, definite identification of enterococci at species level is mandatory to assess their variable sensitivity pattern and to treat accordingly.
Conclusions
In our study, the most frequent species of Enterococcus was E. faecalis, of which a large proportion revealed high level resistance to gentamycin. So, infections with E. faecalis can be especially troublesome to treat because of their frequent resistance to multiple antibiotics, including vancomycin, a drug of last resort for many Gram-positive infections(29).Taking into account their fast capacity of acquiring antibiotic resistance, enterococci may become the most frequent isolated bacteria in hospitalized patients.
Bibliografie
- Facklam RR, Teixeira LM. Topley & Wilson’s Microbiology and Microbial Infections, Lollier L, Balows A, and Sussman M, Eds., "Enterococcus". Oxford University Press, New York, NY, USA, 9th edition, 1998; 669–682.
- Edwards DD. Enterococci attract attention of concerned microbiologists. ASM News. 2000(66):540-5
- Schaberg DR, Culver DH, Gaynes RP. Major trends in the microbial etiology of nosocomial infection. Am J Med. 1991; 91(3B): S72–75
- Ronald AR, Pattulo MS. The natural history of urinary infection in adults. Med Clin North Am 1991; (75):299-312.
- Low DE, Keller N, Barth A, Jones RN. Clinical prevalence, antimicrobial susceptibility, and geographic resistance patterns of enterococci: results from the SENTRY Antimicrobial Surveillance Program, 1997-1999. Clin Infect Dis 2001; 32(2):Suppl:33-45.
- Tyrrell GJ, Turnbull L, Teixeira LM, et al. Enterococcus gilvus sp. nov. and Enterococcus pallens sp. nov. isolated from human clinical specimens. J Clin Microbiol 2002; 40:1140-5.
- Murray BE. The life and times of the enterococcus. Clin Microbiol Rev. 1990; 3:45-65.
- Sinjee S. Vancomycin resistant enterococci faecium isolated from canine urinary tract infections. JCM. 2002; 4659-65.
- Geraci JE, Martin WJ. Antibiotic therapy of bacterial endocarditis. VI. Subacute enterococcal endocarditis; clinical, pathologic and therapeutic consideration of 33 cases. Circulation. 1954; 10:173-94.
- Murray BE. Vancomycin-resistant enterococcal infections. N Engl J Med. 1999; 342(Suppl 10):710-21.
- Cheesbrough M. Microbiology II. Medical laboratory manual for tropical countries. Cambridge University Press, 2000.
- Facklam RR, Collins MD. Identification of Enterococcus species isolated from human infections by a conventional test scheme. J Clin Microbiol. 1989; 27:731-734.
- Clinical and Laboratory Standards Institute. National Committee for Clinical Laboratory Standards. Performance standards for antimicrobial disk susceptibility test. 26th edition M100S.
- Bauer AW, Kirby WWM, Sherris JC, Turk M. Antibiotic susceptibility testing by standardized single disc method. Am J Clin Pathol. 1966; 45:493-6.
- Olawale KO, Fadiora SO, Taiwo SS. Prevalence of Hospital-Acquired Enterococci Infections in Two Primary-Care Hospitals in Osogbo, Southwestern Nigeria. Afr. J. Infect. Dis. 2011; 5(2):40-6.
- Desai PJ, Pandit D, Mathur M, Gogate A. The prevalence, identification and the distribution of various species of enterococci which were isolated from clinical samples, with special reference to the urinary tract infections in catheterized patients. India J Med Microbial 2001; 19:132-7.
- Jain S, Kumar A, Kashyap B, Kaur RI. The clinico-epidemiological profile and the highlevel aminoglycoside resistance in enterococcal septicemia at a tertiary care hospital in east Delhi. Int J App Basic Med Res 2011; 1(2):80-3.
- Facklam RR, Sahm DF, Teixeira LM. Manual of Clinical Microbiology, 7th ed. Enterococcus. 1999; 297-305. ASM Press, Washington, DC, USA.
- Brooks GF, Carroll KC. Jawetz, Melnick & Adelberg’s Medical Microbiology, Streptococcus. 25th edition, Mc Graw Hill, 2010, 206.
- Sreeja S, Sreenivasa Babu PR, Prathab AG. The prevalence and characterization of the Enterococcus species from various clinical samples in a tertiary care hospital. J Clin Diagn Res. 2012; 6(9):1486-8.
- Kapoor L, Randhawa VS, Deb M. Antimicrobial resistance of enterococcal blood isolates at a pediatric care hospital in India. J Infect Dis. 2005; 58:101-3.
- Prakash VP, Rao SR, Parija SC. Emergence of unusual species of enterococci causing infections, South India. BMC Infectious Diseases. 2005; 5:14, http://www.biomedcentral.com/1471-2334/5/14
- Courvalin P. Vancomycin resistance in Gram-positive cocci. Clin Infect Dis 2006; 42(Suppl 1):S25-34.
- Morrison AJ Jr, Wenzel RP. Nosocomial urinary tract infections due to enterococcus. Ten years’ experience at a university hospital. Arch Intern Med 1986; 146:1549-51.
- Lemoine L, Hunter PR. Enterococcal urinary tract infections in a teaching hospital. Eur J Clin Microbiol 1987; 6:574-5.
- Grude N, Tveten Y, Kristiansen BE. Urinary tract infection in Norway: bacterial etiology and susceptibility. A retrospective study of clinical isolates. Clin Microbiol Infect 2001; 7:543-7.
- Orrett FA, Connors E. Enterococcal urinary tract infections: eight years experience at a regional hospital in Trinidad, West Indies. Chin Med J. 2001; 114:90-2.
- Cornia PB, Takahashi TA, Lipsky BA. The microbiology of bacteriuria in men: a 5-year study at a Veterans’ Affairs hospital. Diagn Microbiol Infect Dis. 2006; 56:25-30.
- Huycke MM, Sahm DF, Gilmore MS. Multiple-drug resistant enterococci: the nature of the problem and an agenda for the future. Emerg. Infect. Dis. 1998; 4:239-49.
- Kristich CJ, Rice LB, Arias CA. "Enterococcal Infection - Treatment and Antibiotic Resistance". 2014 Feb 6. In: Gilmore MS, Clewell DB, Ike Y, et al., editors. Enterococci: From Commensals to Leading Causes of Drug Resistant Infection. Boston: Massachusetts Eye and Ear Infirmary.