Fetal goitre secondary to hypothyroidism is the leading cause of intellectual disability. Despite its rarity, many cases of fetal goitre are diagnosed and require prenatal management. We present a comprehensive review about the prenatal diagnosis and treatment of fetal hypothyroid goitre. Fetal thyroid size can be measured by transvaginal ultrasonography from 14 weeks, and by transabdominal ultrasonography from 18 weeks of gestation. The size of the fetal thyroid depends on gestational age. Because fetal goitre can lead to intrauterine, perinatal and obstetrical complications, the optimal treatment is required. Fetal complications are: fetal growth restriction, polyhydramnios, pleural effusion, preterm delivery, craniosynostosis, neurodevelopmental disorders and neonatal asphyxia or fetal death. The optimal intraamniotic L-thiroxine treatment did not normalize the thyroid hormonal levels, but these infants have normal neuromotor and psychoemotional development.
Diagnosticul antenatal şi tratamentul guşei fetale hipotiroidiene
Antenatal diagnosis and treatment of fetal goitrous hypothyroidism
First published: 09 aprilie 2021
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ORL.51.2.2021.4949
Abstract
Rezumat
Guşa fetală secundară hipotiroidismului este principala cauză a dizabilităţii intelectuale. În ciuda rarităţii sale, multe cazuri de guşă fetală sunt diagnosticate şi necesită tratament prenatal. Scopul acestui articol este să prezentăm o revizuire cuprinzătoare despre diagnosticul prenatal şi tratamentul guşei hipotiroidiene fetale. Mărimea tiroidei fetale poate fi măsurată prin ultrasonografie transvaginală de la 14 săptămâni şi prin ultrasonografie transabdominală de la 18 săptămâni de gestaţie. Mărimea tiroidei fetale depinde de vârsta gestaţională. Deoarece guşa fetală poate conduce la complicaţii intrauterine, perinatale şi obstetricale, este necesar un tratament optim. Complicaţiile fetale sunt: restricţia de creştere intrauterină fetală, polihidramnios, revărsat pleural, naşterea prematură, craniosinostoză, tulburări de neurodezvoltare şi de asfixie neonatală sau moarte fetală. Deşi tratamentul optim cu L-thiroxină intraamniotic nu a normalizat nivelurile hormonale tiroidiene, aceşti sugari au o dezvoltare neuromotorie şi psihoemoţională normală.
Fetal goitre is a very rare disease that can be secondary to thyroid dysfunction, generally hypothyroidism. Before Down syndrome cause, congenital hypothyroidism (CH) is the most frequent congenital cause of intellectual disability(1).
The incidence of fetal goitre associated with hypothyroidism is 1/30,000 to 1/50,000 live births; this becomes more frequent in pregnant women with Graves’ disease or other thyroid dysfunctions(2).
Fetal goitre represents an enlargement of the fetal thyroid gland; it may be preventable. This pathology has a predilection for females, with the fetal incidence being 2:1 for females than males(3).
Fetal goitre indicates a thyroid dysfunction, and it can be associated with all types of different thyroid hormone levels: hypothyroidism, hyperthyroidism and even euthyroidism(4,5).
The fetal goitre is a sign of maternal hyperthyroidism when there is a placental passage of TSI (thyroid stimulating immunoglobulins) from the mother with Graves’ disease to the fetus, or a sign of hypothyroidism due to the antithyroid medication given to the pregnant woman(6,7).
While the fetal thyroid develops between the 7th and the 12th weeks of gestation, it reaches its final site in the neck and it does not function autonomously until around the 18-20th weeks. Until that gestational age, having a balanced status of the maternal thyroid hormones is important for the neuromotor and intellectual physiological development of the fetus(8).
These findings may justify that an endocrinological evaluation of the thyroid gland may be highly recommended preconceptionally and during pregnancy, especially in endemic areas. Any pathologies discovered de novo could be diagnosed and treated. Alternatively, the treatment doses can be adjusted for the preexisting pathologies to prevent consecutive fetal thyroid pathology development.
Diagnostic
The first case of antenatal diagnosis of fetal goitre was reported back in 1980, and this was also the first case of fetal hypothyroidism treated in utero with thyroxine, which helped reduce the goitre volume after its administration considerably(9).
Fetal goitre is diagnosed by prenatal ultrasound or, in special cases, combined with fetal magnetic resonance imaging (MRI).
Fetal thyroid size can be measured by transvaginal ultrasonography from 14 weeks, and by transabdominal ultrasonography from 18 weeks of gestation(10).
Many anatomical studies have reported normal length values: first trimester – 3.47 ± 0.54 mm; second trimester – 7.48 ± 1.74 mm; third trimester – 13.66 ± 2.05 mm; at 40 weeks – 21.34 ± 3 mm(11,12).
There are few ultrasound reference curves for fetal thyroid measurements, including fetal thyroid length in a single plane, with measurements of circumference, total transverse diameter or thyroid area and volume(13-19). Barbosa et al. have published very good reference ranges for thiroid measurement(20).
Fetal thyroid examination is not systematically recommended during prenatal screening evaluation. Fetal goitre appears as a hyperechogenic, symmetric mass in the neck region, anteriorly, with a length diameter above the 90-95th percentile for gestational age(16).
Attempts have been made to establish the standard diagnostic criteria by using ultrasonography starting with week 20, but protocols have not yet been developed for this investigation.
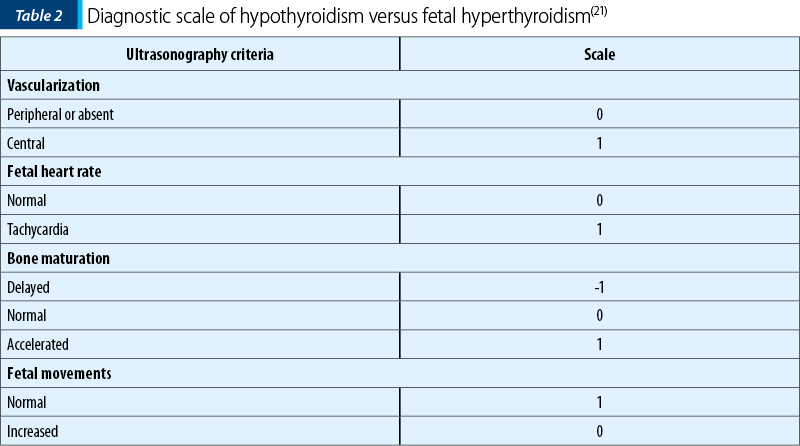
Huel et al. have recommended the analysis of the following four parameters for the diagnosis of hypo- or fetal thyroid hyperfunction: vascular pattern of the goitre, fetal heartbeat, bone maturation of the fetus, fetal movement (Table 1)(21).
Bone maturation was evaluated at 32 weeks using ultrasound. Advanced bone maturation was defined by the presence of the distal ossification centre before 31 weeks and delayed bone maturation as the absence of the distal femoral ossification centre after 33 weeks.
Fetal tachycardia was defined as a fetal heart rate frequency higher than 160 bpm(21).
Also, a useful score scale for the diagnosis fetal hypo- or hyperthyroidism was imagined (Table 2).
Thus, having a total score above 2 suggests the presence of hyperthyroidism, while a score below 2 indicates fetal hypothyroidism.
Fetal MRI is complementary to ultrasonography; it can help in the differential diagnosis and for obtaining a detailed fetal thyroid functionality(22).
After the imaging investigations, the second step involves performing laboratory investigations. It was initially thought that fetal thyroid pathologies could be diagnosed based on the level of thyroid hormones in the amniotic fluid. However, it has been shown that fetal blood collected from the umbilical cord has a much higher sensitivity(7), given that the level of thyroxine in the amniotic fluid is both from fetal and maternal sources. On the other hand, TSH in the amniotic fluid seems to only reflect the fetal level’s, given that it cannot cross the fetal-placental barrier. Cordocentesis is considered the gold standard for diagnosis, although the complication rate is significantly higher, and the technique is significantly more laborious than amniocentesis(23,24).
The etiology of fetal hypothyroidism is diverse; in about 85% of cases, it is caused by thyroid dysgenesis (agenesis, hypoplasia or ectopy). The other 15% of cases of primary congenital hypothyroidism are due to defective hormone synthesis (thyroid dyshormonogenesis) caused by different mutations involving SLC26A4, DUOX2, DUOXA2, DEHAL1, sodium/iodine transporter, thyroid peroxidase (TPO) and thyroglobulin (TG)(25-27).
One particular case where the antepartum diagnosis cannot be made is when there is a congenital resistance to TSH, an autosomal recessive inheritance of TSH resistance caused by inactivating mutations in the TSHR(28). The clinical and paraclinical aspects of these pathologies are extremely varied and are generally associated with normal or low glandular volume, ranging from asymptomatic forms with elevated TSH to forms with a lack of thyroid gland(21,29).
It is also vital to monitor fetuses with Down syndrome, who frequently have low levels of T4 and high levels of TSH, thus associating various forms in terms of severity of primary congenital hypothyroidism. In this way, some of the neurological and psychomotor disorders of newborns with trisomy 21 can be explained by these disorders in the thyroid hormone levels. Their adverse effects on fetal development could be interrupted by the administration of thyroxine in pregnant women with fetuses with specific changes in Down syndrome(30,31).
Complications
Fetal goitre can lead to numerous complications in utero or perinatally, such as fetal growth restriction, polyhydramnios, pleural effusion, preterm delivery, neonatal asphyxia or fetal death, labor dystocia, craniosynostosis, but also the most frequent neurodevelopmental deficits(32,33). Moreover, fetal thyrotoxicosis can lead to tachycardia, cardiac failure or hydrops, and to premature birth or fetal death(34).
The goitre mass can lead to different complications, such as airway compression, polyhydramnios due to compression of the cervical mass on the esophagus, vicious obstetrical presentations due to cervical hyperextension, and numerous childbirth impediments that can lead to neonatal asphyxia and even death.
In addition, due to the cervical mass, neonatal intubation and ventilation are often performed with difficulty(35).
Maternal complications include placental abruption, preterm labor, preeclampsia, congestive heart failure and thyroid storm(36).
Treatment
Deciding on the prenatal treatment represents another challenge because the trade-offs of initiating intrauterine treatment must be balanced with the trade-offs of the “watchful waiting” strategy. The reduction in the thyroid goitre (as observed on ultrasound imaging) is not equivalent to normalising fetal thyroid hormone concentrations.This decision to treat or not is affected by the substance being administered as treatment, generally indicating LT4 or a combination of LT4 and T3, dose, method of administration and frequency. Often, despite treatment, infants are born with hypothyroidism, but with normal neuromotor and psychoemotional development.
In terms of methods of administration, intraamniotic, intramuscular or fetal administration are currently practised. Intraamniotic L-thyroxine hormones administration was proven to be the most effective, although in 9.7% of cases this method led to preterm delivery or chorioamniotitis(37-39). Its efficiency is explained by the deficiency in the transplacental transport of the thyroid hormone.
Doses can vary between 150 and 800 µg, and they are being adjusted depending on the response to the treatment, every few days or a few weeks to a month. The length of time between the last dose of treatment and birth seems to influence the evolution of this pathology in extrauterine life(1).
In cases of fetal hyperthyroidism, a decrease in the dose of maternal antithyroid medication is recommended.
Regarding the way of birth, if the goitre is not a voluminous one, it is recommended to give birth vaginally. In the rest of cases, or in those that require an in utero intervention for airway clearance, giving birth by caesarean section is more suitable(40-43).
There are no guidelines or general recommendations for optimal treatment regimen. However, there are currently a wide variety of approaches to treatment.
In conclusion, the diagnosis and treatment of fetal hypothyroidism goitre are challenging, especially when maternal thyroid function is normal.
Bibliografie
-
Aubry G, Pontvianne M, Chesnais M, Weingertner AS, Guerra F, Favre R. Prenatal Diagnosis of fetal goitrous hypothyroidism in a euthyroid mother: A management challenge. J Ultrasound Med. 2017;36(11):2387-92.
-
Fisher DA, Klein AH. Thyroid development and disorders of thyroid function in the newborn. N Engl J Med. 1981;304:702–12.
-
Stoppa-Vaucher S, Van Vliet G, Deladoëy J. Variation by ethnicity in the prevalence of congenital hypothyroidism due to thyroid dysgenesis. Thyroid. 2011;21(1):13-8.
-
Munoz JL. Fetal thyroid disorders: Pathophysiology, diagnosis and therapeutic approaches. J Gynecolog Obstet Hum Reprod. 2019;48(4):231-3.
-
Ferianec V, Papcun P, Grochal F, Schenková K, Bártová M. Prenatal diagnosis and successful intrauterine treatment of severe congenital hypothyroidism associated with fetal goiter. J Obstet Gynaecol Res. 2017;43(1):232-7.
-
Luton D, Le Gac I, Vuillard E, Castanet M, Guibourdenche J, Noel M, Toubert ME, Léger J, Boissinot C, Schlageter MH, Garel C, Tébeka B, Oury JF, Czernichow P, Polak M. Management of Graves’ disease during pregnancy: the key role of fetal thyroid gland monitoring. J Clin Endocrinol Metab. 2005;90(11):6093-8.
-
Volumenie JL, Polak M, Guibourdenche J, Oury JF, Vuillard E, Sibony O, Reyal F, Raccah-Tebeka B, Boissinot C, Madec AM, Orgiazzi J, Toubert ME, Leger J, Blot P, Luton D. Management of fetal thyroid goitres: a report of 11 cases in a single perinatal unit. Prenat Diagn. 2000;20(10):799-806.
-
Polak M, Luton D. Fetal thyroidology. Best Pract Res Clin Endocrinol Metab. 2014;28(2):161-73.
-
Weiner S, Scharf JI, Bolognese RJ, Librizzi RJ. Antenatal diagnosis and treatment of a fetal goiter. J Reprod Med. 1980;24(1):39-42.
-
Cohen O, Pinhas-Hamiel O, Sivan E, Dolitski M, Lipitz S, Achiron R. Serial in utero ultrasonographic measurements of the fetal thyroid: a new complementary tool in the management of maternal hyperthyroidism in pregnancy. Prenat Diagn. 2003;23(9):740-2.
-
Ozguner G, Sulak O. Size and location of thyroid gland in the fetal period. Surg Radiol Anat. 2014;36(4):359-67.
-
Cicekcibasi AE, Salbacak A, Seker M, Ziylan T, Tuncer I, Buyukmumcu M Developmental variations and clinical importance of the fetal thyroid gland. Saudi Med J. 2007; 28(4):524–8.
-
Bromley B, Frigoletto FD, Cramer D, Osathanondh R, Benacerraf BR. The fetal thyroid: normal and abnormal sonographic measurements. J Ultrasound Med. 1992;11(1):25–8.
-
Ho SSY, Metreweli C. Normal fetal thyroid volume. Ultrasound Obstet Gynecol. 1998;11(2):118–22.
-
Achiron R, Rotstein Z, Lipitz S, Karasik A, Seidman DS. The development of the foetal thyroid: in utero ultrasonographic measurements. Clinical Endocrinol. 1998;48(3):259–64.
-
Ranzini AC, Ananth CV, Smulian JC, Kung M, Limbachia A, Vintzileos AM. Ultrasonography of the fetal thyroid: nomograms based on biparietal diameter and gestational age. J Ultrasound Med. 2001;20(6):613–7.
-
Radaelli T, Cetin I, Zamperini P, Ferrazzi E, Pardi G. Intrauterine growth of normal thyroid. Gynecol Endocrinol. 2002;16(6):427–30.
-
Bernardes LS, Ruano R, Sapienza AD, Maganha CA, Zugaib M. Nomograms of fetal thyroid measurements estimated by 2-dimensional sonography. J Clinical Ultrasound. 2008;36(4):193–9.
-
Gietka-Czernel M, Dębska M, Kretowicz P, Dębski R, Zgliczyński W. Fetal thyroid in two-dimensional ultrasonography: nomograms according to gestational age and biparietal diameter. European J Obstet Gynecol Reprod Biol. 2012;162(2):131–8.
-
Barbosa RM, Andrade KC, Silveira C, Almeida CM, Souza RT, Oliveira PF, Cecatti JG. Ultrasound measurements of fetal thyroid: reference ranges from a cohort of low-risk pregnant women. Biomed Res Int. 2019;2019:9524378.
-
Huel C, Guibourdenche J, Vuillard E, Ouahba J, Piketty M, Oury JF, Luton D. Use of ultrasound to distinguish between fetal hyperthyroidism and hypothyroidism on discovery of a goiter. Ultrasound Obstet Gynecol. 2009;33(4):412-20.
-
Kondoh M, Miyazaki O, Imanishi Y, Hayakawa M, Aikyou M, Doi H. Neonatal goiter with congenital thyroid dysfunction in two infants diagnosed by MRI. Pediatr Radiol. 2004;34(7):570-3.
-
Abuhamad AZ, Fisher DA, Warsof SL, Slotnick RN, Pyle PG, Wu SY, Evans AT. Antenatal diagnosis and treatment of fetal goitrous hypothyroidism: case report and review of the literature. Ultrasound Obstet Gynecol. 1995;6(5):368-71.
-
Grüner C, Kollert A, Wildt L, Dörr HG, Beinder E, Lang N. Intrauterine treatment of fetal goitrous hypothyroidism controlled by determination of thyroid-stimulating hormone in fetal serum. A case report and review of the literature. Fetal Diagn Ther. 2001;16(1):47-51.
-
Figueiredo CM, Falcão I, Vilaverde J, Freitas J, Oliveira MJ, Godinho C, Dores J, Rodrigues MC, Carvalho C, Borges T. Prenatal diagnosis and management of a fetal goiter hypothyroidism due to dyshormonogenesis. Case Rep Endocrinol. 2018;2018:9564737.
-
Vasudevan P, Powell C, Nicholas AK, Scudamore I, Greening J, Park SM, Schoenmakers N. Intrauterine death following intraamniotic triiodothyronine and thyroxine therapy for fetal goitrous hypothyroidism associated with polyhydramnios and caused by a thyroglobulin mutation. Endocrinol Diabetes Metab Case Rep. 2017;2017:17-0040.
-
Blumenfeld YJ, Davis A, Milan K, Chueh J, Hudgins L, Barth RA, Hintz SR. Conservatively managed fetal goiter: an alternative to in utero therapy. Fetal Diagn Ther. 2013;34(3):184-7.
-
Sunthornthepvarakul T, Gottschalk ME, Hayashi Y, Refetoff S. Brief report: resistance to thyrotropin caused by mutations in the thyrotropin-receptor gene. N Engl J Med. 1995;332(3):155-60.
-
Sura-Trueba S, Aumas C, Carre A, Durif S, Leger J, Polak M, de Roux N. An inactivating mutation within the first extracellular loop of the thyrotropin receptor impedes normal posttranslational maturation of the extracellular domain. Endocrinology. 2009;150(2):1043-50.
-
Luton D, Azria E, Polak M, Carré A, Vuillard E, Delezoide AL, Guibourdenche J. Thyroid function in fetuses with down syndrome. Horm Res Paediatr. 2012;78(2):88-93.
-
van Trotsenburg AS, Vulsma T, van Santen HM, Cheung W, de Vijlder JJ. Lower neonatal screening thyroxine concentrations in down syndrome newborns.
-
J Clin Endocrinol Metab. 2003;88(4):1512-5.
-
Mastrolia SA, Mandola A, Mazor M, Hershkovitz R, Mesner O, Beer-Weisel R, Besser L, Shelef I, Loewenthal N, Golan A, Gruzman I, Erez O. Antenatal diagnosis and treatment of hypothyroid fetal goiter in an euthyroid mother: a case report and review of literature. J Matern Fetal Neonatal Med. 2015;28(18):2214-20.
-
Daneman D, Howard NJ. Neonatal thyrotoxicosis: intellectual impairment and craniosynostosis in later years. J Pediatr. 1980;97(2):257-9.
-
Ribault V, Castanet M, Bertrand AM, Guibourdenche J, Vuillard E, Luton D, Polak M; French Fetal Goiter Study Group. Experience with intraamniotic thyroxine treatment in nonimmune fetal goitrous hypothyroidism in 12 cases. J Clin Endocrinol Metab. 2009;94(10):3731-9.
-
Kriplani A, Buckshee K, Bhargava VL, Takkar D, Ammini AC. Maternal and perinatal outcome in thyrotoxicosis complicating pregnancy. Eur J Obstet Gynecol Reprod Biol. 1994;54(3):159-63.
-
Iijima S. Current knowledge about the in utero and peripartum management of fetal goiter associated with maternal Graves’ disease. Eur J Obstet Gynecol Reprod Biol X. 2019;3:100027.
-
Davidson KM, Richards DS, Schatz DA, Fisher DA. Successful in utero treatment of fetal goiter and hypothyroidism. N Engl J Med. 1991;324(8):543-6.
-
Khamisi S, Lindgren P, Karlsson FA. A rare case of dyshormonogenetic fetal goiter responding to intra-amniotic thyroxine injections. Eur Thyroid J. 2014;3(1):51-6.
-
Stewart CJ, Constantatos S, Joolay Y, Muller L. In utero treatment of fetal goitrous hypothyroidism in a euthyroid mother: a case report. J Clin Ultrasound. 2012;40(9):603-6.
-
Nemescu D, Tănasă IA, Stoian DL, Navolan DB, Vinturache AE. Conservative in utero treatment of fetal dyshormonogenetic goiter with levothyroxine, a systematic literature review. Exp Therap Med. 2020; 20:2434-8.
-
Abraham RJ, Sau A, Maxwell D: A review of the EXIT (ex utero intrapartum treatment) procedure. J Obstet Gynaecol. 2010;30:1–5.
-
Chmait RH, Chon AH, Anselmo D, Vanderbilt DL, Townsend J, Julian-Wang B, Don D. In utero fetal intubation for a large neck mass: A minimally invasive EXIT option. Fetal Diagn Ther. 2019;45(4):275–80.
-
Cruz-Martinez R, Moreno-Alvarez O, Garcia M, Méndez A, Pineda H, Cruz-Martinez MA, Martinez-Morales C. Fetal endoscopic tracheal intubation: A new fetoscopic orocedure to ensure extrauterine tracheal permeability in a case with congenital cervical teratoma. Fetal Diagn Ther. 2015;38(2):154–8.