The principles of reconstructive surgery of the maxillofacial region must ensure a good functional result, but also a cosmetic outcome that recreates the natural lines of the face. A great care must be taken by the surgeon in the case of lip defects following tumor pathology due to their importance in the facial harmony. Although there are various well defined procedures for facial reconstruction, local flaps still play a highly significant role due to the ease of harvesting and the good adaptation at the recipient site. Functional evaluation was undertaken in all patients at 2 weeks, 3 and 6 months postoperative. The recovery is faster than for distant site flaps, and the surgical procedure can be performed on patients with comorbidities.
Reconstrucţia defectelor faciale post-rezecţie tumorală cu lambouri locale
Reconstruction of facial defects after tumor resection with local flaps
First published: 07 iunie 2017
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Orl.35.2.2017.798
Abstract
Rezumat
Reconstrucţia plastică a defectelor post-exereză tumorală în sfera orofacială trebuie să asigure atât reintegrarea funcţională a pacienţilor, cât şi un aspect fizionomic cât mai natural. Un interes deosebit îl reprezintă plastia defectelor după ablaţii tumorale extinse ale buzelor, datorită importanţei lor în contextul armoniei faciale. Deşi există numeroase posibilităţi de plastie, lambourile locoregionale reprezintă una dintre cele mai bune variante de refacere a continuităţii ţesuturilor atât datorită accesibilităţii chirurgicale, cât şi integrării rapide şi optime la nivelul situsului receptor. Evaluarea funcţională a pacienţilor s-a efectuat la 2 săptămâni, la 3 luni şi la 6 luni postoperator. Recuperarea pacienţilor este accelerată comparativ cu lambourile de la distanţă şi, de asemenea, intervenţia chirurgicală comportă mai puţine riscuri faţă de pacient.
Introduction
Facial reconstructive surgery can pose serious problems even for a skilled surgeon. When dealing with malignant tumors, the excision surgery must ensure negative margins all around the tumor process. This usually leaves a significant defect that must be addressed in the same session as the tumor surgery. Many times, these types of defects are composed of soft tissue (skin, muscle, fascia, nerves and vessels) and also hard tissues. The best treatment is provided through the use of free flaps which can ensure a success rate of more than 95%(1). But not all the patients are good candidates for this high demanding procedure and often the surgeon must take into consideration the medical status of the patient - the age, the social and economic status, the patient’s motivation. The medical team must evaluate the benefit of distant flap harvest in each individual case. Local flaps are preferred whenever they are fit to; because of the ease of the access, they can be performed immediately after the excision surgery, they have the same color and texture as the recipient site, and they don’t require a prolonged healing period.
The defects of the lower lip are managed depending on their size with flaps from:
- the remaining lip
- the upper lip
- the cheek region.
Depending on their size, the defects can be classified as(2):
A. Less than 30% - primary closure with V, W excision.
B. 30% to 50% - lip sharing technics (Abbe-Estlander, Johanson, Karapandzic, McGregor).
C. More than 50% - Camille Bernard.
D. Total lip reconstruction - bilateral cheek advancement, bilateral nasolabial flaps.
The ear is a site that is usually affected by base cell carcinoma rather than squamous cell, but there are cases of tenebrous long-lasting malignant tumor which can lead to important tissue sacrifice. Even there are few muscle attachments in the region, a great care must be taken when reconstructing the superficial tissues and also the underlying cartilage.
Depending on the size of the defect, there are various choices for reconstruction: primary closure, second intention healing, skin graft, island transposition flap, tubed pedicle flap, retro auricular advancement flap, and for larger defects, the cervical advanced flaps.
Nasal reconstruction has developed together with plastic surgery, especially from the 19th century onwards. There are numerous flaps, but they all come from three major regions: the forehead, the cheek and the remaining nose tissues. The most common indication for nasal reconstruction is ablative surgery for basal cell carcinoma. Basic principles should take into consideration(3):
- Replacement of missing tissue. Ideally, the flap should mimic the quality and quantity of the adjacent tissues.
- The cosmetic appearance of the patient. The surgeon should recreate the design and texture of a normal nose.
- The scars should be hidden in the least seen places.
Case presentation
Case I
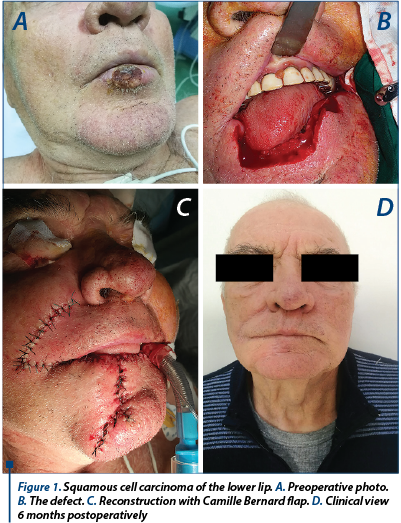
A 70-year-old male patient presented in the ENT and maxillofacial department of “Elias” University Hospital for a lower labial mass. He had noticed a change in the aspect of his lower lip about 2 years ago. At first, there was a small ulceration that did not heal. It was covered by a crust that would sometimes detached itself leaving a bleeding site. The ulceration turned slowly into a proliferative process that grew to the present size. The patient did not seek any treatment for his condition.
An ulcerative-vegetative tumor mass was noticed at the level of the right side lower lip, that exceeded the midline and was affecting the mucosal edge of the lip. The tumor was approximate round, 2/2 cm in size, with irregular margins, covered by hemorrhagic crusts that left a bleeding site when detached. The patient did not complain of any pain. In his lower left lip, there was a lesion of actinic cheilitis. There was no lymph node involvement.
The patient was a farm worker, had been smoking for 30 years, and declined alcohol use. He had been admitted to the hospital 5 years ago with a cerebral stroke for which he took antiaggregant medication.
A decision was made to surgically treat the lesion, resect also the vermillion affected by the cheilitis and reconstruct the defect in the same session with a Camille Bernard right flap.
There was no complication, and the histology report confirmed the clinical diagnosis: T2NoMx G2 squamous cell carcinoma. At 6-month follow-up, there were no relapses.
Case II
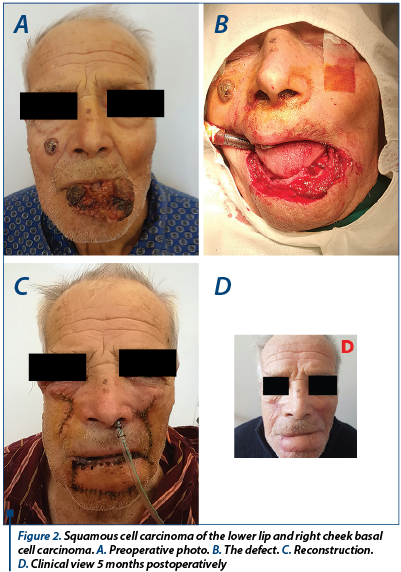
An 82-year-old male patient was admitted to the ENT and maxillofacial department of “Elias” University Hospital with a lower lip tumor mass. The process started approximately 5 years ago as a small ulceration that did not heal and slowly grew to its current size. The patient did not follow any treatment. He had class II hypertension controlled by specific medication and underwent two general anesthesia operations: appendicectomy and inguinal hernia.
The tumor presented as an ulcerated exophytic mass that affected the patient’s whole lower lip; it was covered by hemorrhagic crusts - the patient did not accuse any pain.
There was a secondary tumor on his right cheek covered by hemorrhagic crusts, 2 cm in size. The second tumor appeared approximately 2 years before the presentation. This process affected only the skin tissue and did not appear to progress into the underlying muscle. There was no lymph node involvement.
The patient was a retired farm worker, with a lot of sun exposure, had been smoking 20 cigarettes per day for 50 years, and was a social drinker with no history of hard liquor abuse.
The patient underwent excision surgery of the lower lip and reconstruction with a pedicle nasolabial flap. The right cheek tumor was also resected and a primary closure was performed.
The histology report concluded that the lip tumor was a T3NxMx G2 squamous cell carcinoma and the cheek mass was a T1NxMx basal cell carcinoma with negative margins. The healing was successful, with good functionality of the oral cavity.
Case III
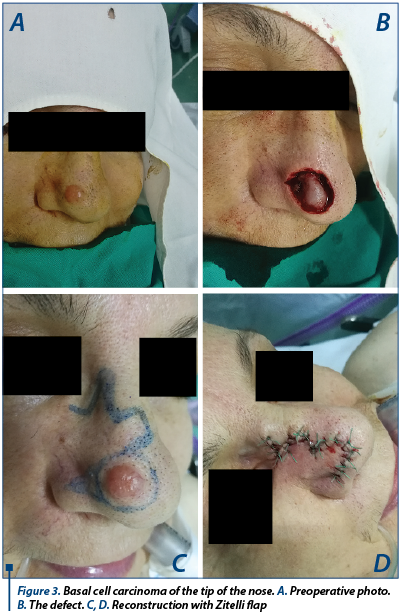
A 45-year-old female patient presented with a nodal tumor proliferation on the tip of the nose; the patient stated that the disease started 3 years ago as a small node and grew slowly in size. The tumor was approximate round, 15 mm in diameter, with irregular margins, solid at palpation. The patient had no medical history. An elliptic excision was planned, followed by a Zitelli bilobed flap. Healing went uneventful. The histology report was basal cell carcinoma.
Case IV
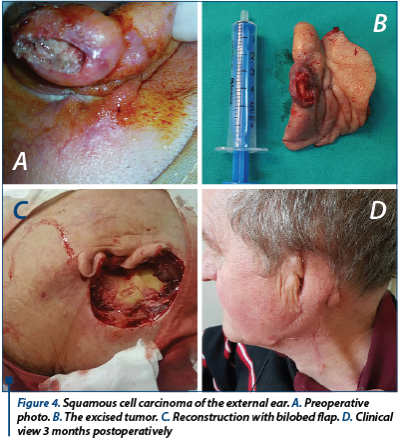
A 58-year-old male patient presented at the ENT and maxillofacial department of “Elias” University Hospital with a terebrant process of the left ear. The patient previously underwent two local excision in another department. It first started as an ulceration 6 years ago, and had been surgically resected 2 years before the presentation. The tumor was an ulcerated endophytic process that affected the posterior side of the external ear, but did not progress into the external auditory canal and did not involve the mastoid process. There was no cervical node involvement. The patient was an electrician, a heavy smoker, declined alcohol abuse, did not take any medication and had no other comorbidities.
The external ear was resected, sparing only the ear lobe. The reconstruction was carried out with the use of a bilobed cervical advanced flap. The histological examination was conclusive for T3NxMx squamous cell carcinoma with no bone involvement. The healing went uneventful.
Discussion
Ablative facial surgery can cause not only functional, but also cosmetic impairment, reducing the chance of a rapid social adaptation. The defects can be left to granulate by secondary healing, reconstructed with local and regional flaps or when there is a composite tissue loss with microsurgical free flaps.
The local flaps have many advantages(4):
- Ease of surgical access.
- The same texture, color and thickness as the recipient site.
- Optimal healing with minimal hospitalization.
Flap survival depends on the vascular pedicles at the base of the flaps and the newly formed anastomosis from the recipient site to the flap.
There are many classifications of the local flaps depending on their shape (rhombic, bilobed) or method of delivery to the recipient site (rotational, advancement, pivotal, transposition). There is not a perfect flap for every case and each reconstruction procedure should be carefully chosen to provide a good functional and esthetic outcome, taking into consideration the patient’s needs and comorbidities. Usually, when reconstructing a facial defect, the flaps are not singular specific units, but rather a combination.
The nose, lying in the center of the face, can be very demanding in reconstructive surgery because of the visible scar lines. Defects of the upper two third can be reconstructed with thin skin grafts; another good option is the dorsal nasal flap. For the lower third usually bilobed nasal flap (Zitelli) is a good choice(5). When the tip of the nose is missing, the best alternative in plastic surgery is the use of forehead flap, usually from the ipsilateral side. This type of flap is limited in length by the hairline.
Lip malignancy is the most frequent cancer of the oral cavity and a very high percentage of this is squamous cell carcinoma(6). There is a predominance of males rather than females, and is correlated with tobacco and alcohol usage(7). The lip reconstruction should focus on the functional rehabilitation of the oral cavity. Frequently, there is only lower lip involvement. Depending on the size of the defect, a simple primary closure can be performed with an advancement of the oral labial mucosa. For full thickness defects, pedicle lip-switch flaps represent a very good choice. The pedicle must be divided usually after 3 weeks. The main disadvantage is some degree of microstomia. For larger defects, that cross the midline, adjacent cheek flaps provide good support. For defects involving the whole lip, bilateral flaps can be used. The mucosa should be incised superior to the muscle incision, in order to ease the reconstruction of the vermilion. The most important aspects for the preservation of function in lower lip reconstruction are labial competence, preservation of sensation and unobstructed muscle activity(8).
The auricular reconstruction following malignant tumor excision represents a great challenge for facial plastic surgery and requires a good understanding of the various techniques, in order to allow for the best outcome for the patient. Approximately 1/10.000 patients with external ear pathology is diagnosed with a malignant tumor(9). The tumor can have various origins, such as the skin, cartilage or bone. The most common malignancies are squamous and basal cell carcinomas. Depending on the size of the tumor, the defect can have different sizes and shapes that necessitate various reconstruction technics. Small defects can be dealt with wedge excision and primary closure. Medium size defects can usually be reconstructed with helical chondrocutaneous advancement flaps(10). Larger defects require cervical advancement flaps, prosthetic rehabilitation, costal grafts, and many times a combination of these.
Conclusions
Although there are incredible advances in the microvascular free flap procedures and also in prosthetic rehabilitation, there are many situation when local flaps provide the best outcome for reconstructive facial surgery.
Bibliografie
2. Siqueira EJ, Alvarez GS, Laitano FF, et al. Lower lip reconstruction strategies. Rev Bras Cir Plast 2012; 27:536-41.
3. Menick, Frederick J. Nasal Reconstruction, Plastic & Reconstructive Surgery, 2010, 125(4):138e-150e.
4. Rao JK, Shende KS. Overview of Local Flaps of the Face for Reconstruction of Cutaneous Malignancies: Single Institutional Experience of Seventy Cases. Journal of Cutaneous and Aesthetic Surgery. 2016; 9(4):220-225. doi:10.4103/0974-2077.197029.
5. Cho M, Kim DW. Modification of the Zitelli Bilobed Flap. A Comparison of Flap Dynamics in Human Cadavers. Arch Facial Plast Surg. 2006; 8(6):404-409. doi:10.1001/archfaci.8.6.404.
6. Jovanovich A, Schulten EAJM, Kostense PJ, Snow GB, van der Waall I. Squamous cell carcinoma of the lip and oral cavity in the Netherlands; an epidemiological study of 740 patients. J Craniomaxillofac Surg 1993; 21:149-52.
7. Maruccia M, Onesti MG, Parisi P, Cigna E, Troccola A, Scuderi N. Lip cancer: a 10-year retrospective epidemiological study, Anticancer Res. 2012 Apr; 32(4):1543-6.
8. Backer SH. Local Flaps in Facial Reconstruction. London: Saunders; 2008:512-5.
9. Nemechek AJ, Amedee RG. Tumors of the external ear, J La State Med Soc. 1995 Jun; 147(6):239-42.
10. Krunic AL, Weitzul S, Taylor RS. Chondrocutaneous advancement flap for reconstruction of helical rim defects in dermatologic surgery, Australas J Dermatol. 2006 Nov; 47(4):296-9.