Adenomyosis is a benign gynecological pathology, commonly found in women aged 40-50 years old. The nonspecific clinical presentation and the lack of standardized diagnostic criteria make this pathology a challenge for clinicians. We present a rare case of adenomyoma in a 73-year-old woman with obesity, cardiovascular pathology and oncological history of nasal squamous cell carcinoma, who presented to our department for postmenopausal bleeding. In this case report, we aim to highlight the diagnostic and therapeutic management for persistent uterine bleeding at an advanced age, discussing the paraclinical evidence guiding the diagnosis. Definitive surgical treatment by total hysterectomy with bilateral salpingo-oophorectomy contributed fundamentally to the diagnosis of adenomyoma. The therapeutic option took into account the persistence of the patient’s symptoms and related comorbidities.
Adenomyosis: a rare differential diagnosis in postmenopause – case report
Adenomioza: un diagnostic diferenţial rar în postmenopauză – prezentare de caz
First published: 30 septembrie 2023
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.41.3.2023.8736
Abstract
Rezumat
Adenomioza este o patologie ginecologică benignă, întâlnită frecvent la femeile cu vârsta cuprinsă între 40 şi 50 de ani. Prezentarea clinică nespecifică şi lipsa criteriilor standardizate de diagnostic fac din această patologie o provocare pentru clinicieni. Prezentăm un caz rar de adenomiom la o femeie de 73 de ani, cu obezitate, patologie cardiovasculară şi antecedente oncologice de carcinom nazal scuamos, care s-a prezentat în departamentul nostru pentru metroragie în postmenopauză. În acest caz, ne propunem să evidenţiem managementul diagnostic şi terapeutic al hemoragiilor uterine persistente la vârsta înaintată, discutând dovezile paraclinice care ghidează diagnosticul. Tratamentul chirurgical definitiv prin histerectomie totală cu anexectomie bilaterală a contribuit fundamental la diagnosticarea unui adenomiom. Opţiunea terapeutică a ţinut cont de persistenţa simptomelor şi de comorbidităţile pacientei.
Introduction
Adenomyosis is a benign gynecological pathology defined by the presence of endometrial glands and stroma in the uterine myometrium, associated with a degree of hyperplasia of the smooth muscles, thus leading to the destruction of the myometrial architecture(1,2).
Histologically, adenomyosis can be classified as localized or diffuse. The localized form, described as a circumscribed nodular formation enclosed by hypertrophic and hyperplastic myometrium, is called adenomyoma. In diffuse adenomyosis, the heterotopic islands of endometrial glands and stroma are scattered, diffusely invading the myometrium.
In terms of epidemiological evidence, the incidence and prevalence of the disease are increased at the reproductive age(3), diagnosed between 40 and 50 years old. It is estimated that the total incidence of adenomyosis is 1.03%, while the prevalence is around 0.8%(4).
The precise pathophysiological processes underlying the onset of the disease are not yet completely comprehended. Over time, several theories have been proposed to explain the mechanism of adenomyosis. Recent data from the literature suggest the involvement of inflammatory processes that result from repetitive microtrauma at the level of endometrium-myometrium junction, retrograde menstrual flow, and metaplasia of myometrial stem cells. At the same time, abnormal hormonal profile, prolactin, estrogen and progesterone levels are strongly involved in pathogenesis. Exposure to various endocrine disruptors also leads to epigenetic changes, with altered gene expression and DNA methylation being able to strongly contribute to the development of this pathology(5,6). The most well-known risk factors for adenomyosis are multiparity, an increased number of abortions and uterine surgery, including caesarean section(2,7). Hyperestrogenism has been associated with an increased risk of adenomyosis, explaining the regression after menopause. Smoking may be considered a protective factor, because it leads to a decrease in plasma estrogen concentrations. At the molecular level, a close correlation was observed between high levels of leukemia inhibitory factor, IL 6, IL 8 and the occurrence of ectopic endometrial tissue in uterine myometrium(5).
The diagnosis of adenomyosis is quite difficult, given the absence of standardized diagnostic criteria. Thus, most often, the diagnosis is made postoperatively, after the morphopathological examination of the hysterectomy specimen. The imaging diagnostic techniques are currently in continuous development, contributing to a better acknowledge of the pathology. The main imagistic methods to evaluate adenomyosis are transvaginal ultrasound and nuclear magnetic resonance imaging. Currently, no imaging diagnostic criteria are described, but there are certain signs and aspects that can guide the clinician to a correct diagnosis(1). Classically, transvaginal ultrasound examination reveals an enlarged uterus with increased volume and regular contour, accompanied by thickened endometrial-myometrial interface. Asymmetry can be observed in the myometrial wall, along with the presence of anechoic microcysts and hyperechoic nodules within the myometrium. When assessing vascularity by Doppler scan, adenomyosis exhibits a distinct linear pattern of blood flow, contrasting with leiomyomas which typically demonstrate peripheral vascularity(6,8,9).
From a clinical point of view, the symptoms are unspecific and can include metrorrhagia, dysmenorrhea, chronic pelvic pain and abundant menstrual flow. Dysmenorrhea and metrorrhagia are described in approximately 65% of cases and, thus, represent the main clinical manifestations of adenomyosis(1,10). Metrorrhagia can be explained by the process of myometrial hypertrophy and all its consequences: angiogenesis, uterine contractions, elevated serum levels of prostaglandins and estrogen. Furthermore, adenomyosis remains asymptomatic in 30% of cases and it often coexists with other gynecological comorbidities that express the same clinical presentation(7). Endometriosis and uterine leiomyomas are the most commonly associated pathologies with adenomyosis. Data from the literature show that in 15-57% of cases patients who are diagnosed with adenomyosis also have leiomyomas(2,11-13).
Case report
We present the case of a 73-year-old patient who was admitted to the Department of Obstetrics and Gynecology of the University Emergency Hospital Bucharest for recurrent postmenopausal metrorrhagia in the last year. From the patient’s medical records, we mention a nasal squamous cell carcinoma treated by surgery and radiotherapy, hypertension, heart failure, dyslipidemia, obesity and recurrent urinary tract infections. The patient also had a history of abdominal surgery: appendectomy and umbilical hernia repair.
Regarding the gynecological history, the patient had a single vaginal birth, no abortions, late menarche, at 16 years old, irregular menstrual cycles with high-flow menstruation and dysmenorrhea, as well as premature menopause, at the age of 38 years old.
The gynecological clinical exam revealed a normal sized uterus, atrophic cervix, non-painful adnexal areas and normal Douglas pouch.
The laboratory tests included complete blood count, coagulation profile and screening for HIV, syphilis, hepatitis B and C. Additionally, samples were collected for vaginal discharges test, cervical culture and Pap smear. The blood results revealed hypochromic microcytic anemia and mild hyperkalemia, while the Pap smear was negative for intraepithelial or malignant lesions. During hospitalization, the patient underwent interdisciplinary evaluation by the cardiology, otolaryngology and dermatology departments.
Chest, abdomen and pelvic computed tomography scan was also performed, revealing a 6.5/5.5 cm uterus with a markedly hypodense intracavitary image and a thin and intense iodophilic endometrium. The ovaries were normal as structure and dimensions, without superficial or deep adenomegaly in the examined segments. Mediastinal millimeter lymph nodes were detected; no secondary pulmonary, hepatic or bone determinations were observed, no fluid was detected at the pericardial, pleural or peritoneal level at the time of examination.
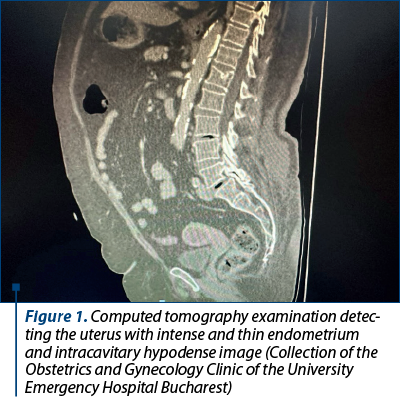
Dilation and curettage (D&C) procedure was initially chosen for both diagnostic and hemostatic purpose. The histopathological examination of endometrial tissue sample showed glandular endometrial fragments without atypia and dilacerate endocervical fragments without further noticeable histopathological changes. Yet, the abnormal uterine bleeding persisted after the D&C procedure.
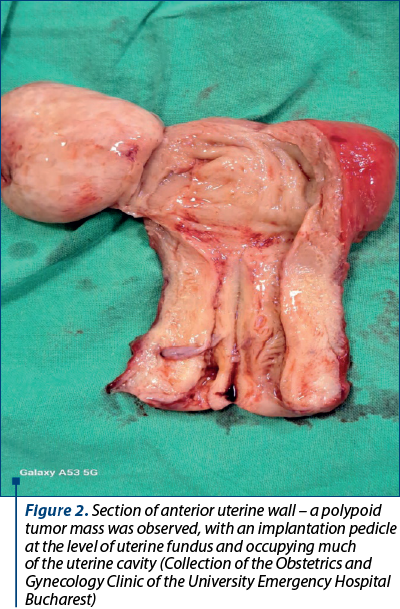
Considering the clinical features, along with the patient’s age and comorbidities, the definitive treatment by total hysterectomy with bilateral salpingo-oophorectomy was decided. Intraoperatively, the normal sized uterus presented increased and inhomogeneous consistency, while the ovaries appeared normal, physiologically involuted. The anterior uterine wall was sectioned (Figure 2) for frozen section procedure that revealed a uterine adenomyoma.
After six hours of follow-up in the intensive care unit (ICU), the postoperative evolution of the patient was uneventful under standard medication, including antibiotics, anticoagulants, analgesics, proton pump inhibitors and probiotics.
The definitive histopathological result on paraffin sections of the specimen was adenomyosis. It described a whitish, nodular tumor with cystic areas, elastic consistence, measuring 4/2.6/2.3 cm. The tumor consisted of a benign proliferation of smooth, uninucleated muscle fibers, arranged around cystic dilated endometrial glands, without cyto-nuclear atypia. It was implanted in the fundic part of the uterus and occupied much of the uterine cavity. Rare intraepithelial micro-abscesses were observed in the exocervical and endocervical epithelium, and the ovaries presented atrophy. Apart from the focal adenomyosis, the endometrium was atrophic, glandular-cystic, slightly dilacerate and had a unitary ratio between the glands and stroma.
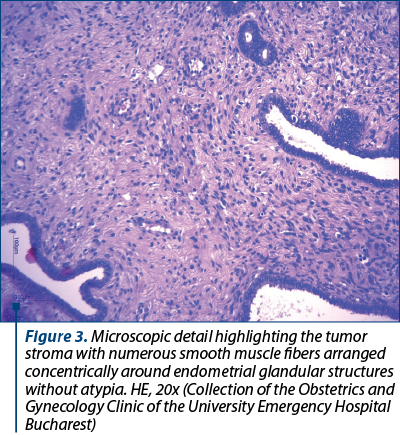
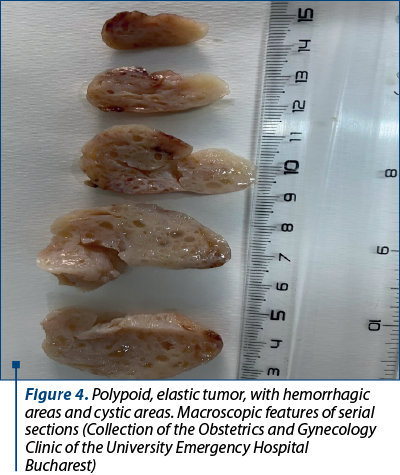
The patient was discharged after four days, in a good overall condition, with a well-balanced cardiac and hemodynamic status, without surgical complications.
Discussion
Postmenopausal bleeding represents a warning sign for any clinician and, regardless of bleeding flow, it should always be further investigated. The incidence and prevalence of postmenopausal bleeding are significantly higher in the early years of menopause and decrease over time, as the temporal distance from the onset of menopause increases(14). The most common causes of postmenopausal bleeding are endometrial atrophy, endometrial polyps, leiomyomas, endometrial neoplasia, hormone therapy and adenomyosis(14-16).
In our case, anamnestic data and clinical examination raised the suspicion of endometrial neoplasia, but it was not histologically confirmed. A meta-analysis of 129 studies, involving 40,790 patients, showed a pooled risk of 9% of endometrial cancer among women with postmenopausal bleeding(17). Adenomyosis is a difficult diagnosis to establish before histopathological examination, since the clinical presentation is nonspecific and, at the same time, there are no standard paraclinical criteria to indicate the diagnosis. Data from various studies evaluating hysterectomy specimens describe histopathological findings of adenomyosis in 10% to 70% of cases(18).
In the presented case, it is important to mention the need for differential diagnosis between adenomyoma and leiomyoma; both can have similar symptomatology, the characteristics observed in imaging examinations are comparable, with a described mass effect on the uterine cavity. The magnetic resonance imaging can help in differentiating the two pathologies by objectifying high-intensity signal-intensity outbreaks on T2-weighted images(6).
The therapeutic strategy for adenomyosis is complex and it is chosen according to the patient’s age, symptoms and desire to maintain fertility. Drug therapy focuses mainly on counteracting the symptoms, such as bleeding and pain(2). The therapeutic management is represented by the administration of progestins, intrauterine devices based on levonorgestrel, danazol and aromatase inhibitors. Danazol has limited utility in adenomyosis, with limited available data; also, androgenic side effects lead to a decrease in the prescription of this medicine(19). Hysterectomy remains the final treatment choice, being the only therapeutic option that completely relieves the symptoms and is especially suitable in patients with a noisy clinical presentation and who are menopausal or do not intend to conceive(1).
In this case, hysterectomy was practiced both for diagnostic and therapeutic purposes, since abnormal bleeding persisted after uterine biopsy. Given the patient’s comorbidities, especially heart failure, chronic bleeding would have led to severe anemia, which could hasten an acute exacerbation of chronic heart failure. Thus, the surgery managed to establish the definitive diagnosis and retrieved a satisfactory quality of life.
Conclusions
Adenomyoma is a rare finding in menopause and requires the exclusion of many differential diagnoses, due to the fact that it can mimic or coexist with a large number of other pathologies. Given the lack of uniform diagnostic criteria, hysterectomy manages to establish a definitive diagnosis for both focal and diffuse adenomyosis.
Conflict of interest: none declared.
financial support: none declared.
This work is permanently accessible online free of charge and published under the CC-BY licence.

Bibliografie
-
Struble J, Reid S, Bedaiwy MA. Adenomyosis: A clinical review of a challenging gynecologic condition. J Minim Invasive Gynecol. 2016;23(2):164–85.
-
Vannuccini S, Petraglia F. Recent advances in understanding and managing adenomyosis. 2019;8:F1000 Faculty Rev-283.
-
Upson K, Missmer SA. Epidemiology of adenomyosis. Semin Reprod Med. 2020;38(2–3):089–107.
-
Yu O, Schulze-Rath R, Grafton J, Hansen K, Scholes D, Reed SD. Adenomyosis incidence, prevalence and treatment: United States population-based study 2006–2015. Am J Obstet Gynecol. 2020;223(1):94.e1-94.e10.
-
Zhai J, Vannuccini S, Petraglia F, Giudice LC. Adenomyosis: mechanisms and pathogenesis. Semin Reprod Med. 2020;38(2–3):129–43.
-
Levy G, Dehaene A, Laurent N, et al. An update on adenomyosis. Diagn Interv Imaging. 2013;94(1):3–25.
-
Chapron C, Vannuccini S, Santulli P, et al. Diagnosing adenomyosis: an integrated clinical and imaging approach. Hum Reprod Update. 2020;26(3):392–411.
-
Andres MP, Borrelli GM, Ribeiro J, Baracat EC, Abrão MS, Kho RM. Transvaginal ultrasound for the diagnosis of adenomyosis: systematic review and meta-analysis. J Minim Invasive Gynecol. 2018;25(2):257–64.
-
Bazot M, Daraï E, Rouger J, Detchev R, Cortez A, Uzan S. Limitations of transvaginal sonography for the diagnosis of adenomyosis, with histopathological correlation. Ultrasound Obstet Gynecol. 2002;20(6):605-11.
-
Bragheto AM, Caserta N, Bahamondes L, Petta CA. Effectiveness of the levonorgestrel-releasing intrauterine system in the treatment of adenomyosis diagnosed and monitored by magnetic resonance imaging. Contraception. 2007;76(3):195–9.
-
Boeer B, Wallwiener M, Rom J, Schoenfisch B, Brucker SY, Taran FA. Differences in the clinical phenotype of adenomyosis and leiomyomas: a retrospective, questionnaire-based study. Arch Gynecol Obstet. 2014;289(6):1235–9.
-
Taran F, Stewart E, Brucker S. Adenomyosis: epidemiology, risk factors, clinical phenotype and surgical and interventional alternatives to hysterectomy. Geburtshilfe Frauenheilkd. 2013;73(09):924–31.
-
Brucker SY, Huebner M, Wallwiener M, et al. Clinical characteristics indicating adenomyosis coexisting with leiomyomas: a retrospective, questionnaire-based study. Fertil Steril. 2014;101(1):237-241.e1.
-
Carugno J. Clinical management of vaginal bleeding in postmenopausal women. Climacteric. 2020;23(4):343–9.
-
Clarke MA, Long BJ, Sherman ME, et al. Risk assessment of endometrial cancer and endometrial intraepithelial neoplasia in women with abnormal bleeding and implications for clinical management algorithms. Am J Obstet Gynecol. 2020;223(4):549.e1-e13.
-
Kim MK, Jung YS, Chon SJ, et al. Common causes of postmenopausal bleeding in Korean women: 10-year outcomes from a single medical center. J Korean Med Sci. 2017;32(5):830-4.
-
Clarke MA, Long BJ, Del Mar Morillo A, Arbyn M, Bakkum-Gamez JN, Wentzensen N. Association of endometrial cancer risk with postmenopausal bleeding in women: a systematic review and meta-analysis. JAMA Intern Med. 2018;178(9):1210-22.
-
Habiba M, Benagiano G. Classifying adenomyosis: Progress and challenges. Int J Environ Res Public Health. 2021;18(23):12386.
-
Pontis A, D’Alterio MN, Pirarba S, de Angelis C, Tinelli R, Angioni S. Adenomyosis: a systematic review of medical treatment. Gynecol Endocrinol. 2016;32(9):696–700.
Articole din ediţiile anterioare
Raport de examinare ginecologică bazat pe consensul grupurilor internaţionale de studiu al tumorilor
Grupul IETA, fondat în 2008 la Chicago, cu ocazia Congresului Mondial de Ultrasonografie în Obstetrică şi Ginecologie, a publicat în ianuarie 2010 ...
Diagnosticul adenomiozei uterine la pacientele de vârstă reproductivă
Adenomioza uterină este o patologie ginecologică ce este caracterizată de prezenţa insulelor ectopice de ţesut endometrial în miometru. Această ...
Adenomioza – algoritm de diagnostic, tratament şi implicaţii obstetricale
Adenomioza continuă să fie o provocare pentru ginecologi, deşi posibilităţile de diagnostic au progresat foarte mult. Tehnicile imagistice moderne ...
Consideraţii de actualitate privind rolul histeroscopiei în diagnosticul şi tratamentul adenomiozei la pacientele infertile
O definiţie histologică clasică pentru adenomioză este invazia miometrului de glande sau stromă endometrială mai mult de 2,5 mm de la nivelul zon...