Fetal and maternal outcomes are related to maternal pre-pregnancy Body Mass Index (BMI) and to the weight gain during pregnancy. Women with overweight or obesity encounter almost a quarter of the complications that develop during pregnancy. The importance of excessive weight gain prophylaxis in pregnancy led to the development of certain guidelines. The Committee on Nutritional Status during Pregnancy and Lactation of the Institute of Medicine (IOM; now, National Academies of Sciences, Engineering and Medicine) stated that, for a favourable outcome of a pregnancy, the weight of the infant should be between 3000 and 4000 grams. The management of the pregnancies should be dictated by the maternal BMI and should be particularized regarding the presence of gestational diabetes mellitus, in which active intervention, including treatment, is needed.
Actualităţi privind greutatea în sarcină
Actualities regarding weight in pregnancy
First published: 20 decembrie 2021
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ObsGin.69.4.2021.5773
Abstract
Rezumat
Rezultatele materne şi fetale ale sarcinii sunt strâns legate de indicele de masă corporală (IMC) matern anterior sarcinii şi de creşterea în greutate în timpul sarcinii. Femeile cu exces ponderal sau cu obezitate înregistrează aproape un sfert din complicaţiile care se dezvoltă în timpul sarcinii. Importanţa profilaxiei creşterii excesive în greutate în timpul sarcinii a condus la elaborarea unor linii directoare. Comitetul pentru starea nutriţională în timpul sarcinii şi alăptării al Institutului de Medicină (IOM; actualmente, Academiile Naţionale de Ştiinţe, Inginerie şi Medicină) a declarat că un rezultat favorabil al sarcinii presupune o greutate a sugarului între 3000 şi 4000 de grame. Gestionarea sarcinilor ar trebui să fie dictată de IMC matern şi ar trebui să fie particularizată în ceea ce priveşte prezenţa diabetului zaharat gestaţional, în care este necesară intervenţia activă, inclusiv tratamentul.
Introduction
The outcomes of the fetal and the maternal evolutions are both in direct relationship with the pre-pregnancy Body Mass Index (BMI) of the mother and also with the weight gain during pregnancy(1). For example, perinatal mortality is at least three times higher in newborns from obese mothers compared to newborns from thin mothers(2).
Females with overweight status or with obesity, reflected by a BMI above 25 kg/m2 and below 30 kg/m2 and, respectively, with a BMI ≥30 kg/m2, have been reported to encounter almost a quarter of the complications that are developed during pregnancy, such as preeclampsia, preterm birth, gestational diabetes mellitus (GDM) or gestational hypertension(3).
On the other hand, pregnant women with obesity which develop a significant weight gain during pregnancy present an even higher risk of complications for the mother, such as postpartum weight retention with maintaining the status of obesity or aggravating the previous status. On the other hand, it is important to highlight the fact that excessive weight gain during pregnancy could be present in underweight (BMI<18.5 kg/m2) and obese women, being exposed to the risk of developing small for gestational age (SGA) fetuses, with short-term and long-term risks(1,3,4). An emphasis should be given to the malformation developed by the fetuses, especially in obese females, where there is a “dose-response” correlation of the appearance of orofacial clefts or limb, genital organs, heart, nervous system, digestive system malformations and the gradually increasing weight class of the mother(5).
The importance of excessive weight gain prophylaxis in pregnancy is a worldwide concern, a fact that led to the development of guidelines in this direction(6).
Obesity before pregnancy
Pre-pregnancy obesity registers an increasing trend, alongside the increment in the frequency of adverse pregnancy outcomes, a fact that determines also the association with high blood pressure(7).
Loss of control over eating is an element that favours the appearance and maintenance of overweight or obesity status, and is important to be a part of the screening in females of reproductive age with higher BMI(8).
A study suggests that there is a small effect of the link between birth anthropometric measurement in case of mothers with pre-pregnancy overweight or obesity status and the overweight or obesity status of the fetus in childhood, with a stronger association in case of the presence of GDM or of hypertensive disorders(9).
Weight gain in pregnancy
Physiologic weight gain normally consists in the fetus weight and the maternal retention of fat and water and is distributed as follows: 0.7 kg for the placenta, 0.9 kg for the amniotic fluid, 3.2 to 3.6 kg for the fetus, 0.9 kg for the uterine hypertrophy, 0.45 to 1.4 kg for the breast enlargement, 2.7 to 3.6 kg of fat stores, 1.4 to 1.8 kg for the increased blood volume, 0.9 to 1.4 kg for the increased extravascular fluid volume, with a total of 11.15 to 14.3 kg(10). The measurement of the weight gain is rarely precisely available, varying, for the initial weight, from the weight from the self-reported or measured periconceptional weight (in case of prenatal care, it is initiated after the first trimester) to the first prenatal visit, and the final weight is the one from the last prenatal visit(10).
A particular situation is represented by the multiple pregnancy, a status of accelerated consumption of nutrients, even to starvation, especially in the second part of a twin pregnancy, a fact that leads to low fasting glucose level associated with a very high insulin response to eating(11). The recommendations of weight gain for multiple pregnancies are especially for twin pregnancies, with a recommendation of a weight gain of 15.87-20.41 kg(12).
The Committee on Nutritional Status During Pregnancy and Lactation of the Institute of Medicine (IOM; now, National Academies of Sciences, Engineering and Medicine), based on an analysis of the published data from the literature, stated that, for a favourable outcome of a pregnancy, the weight for a term liveborn infant should range between 3000 and 4000 grams. Moreover, they concluded that pre-pregnancy BMI is an independent factor than gestational weight and should be used to recommend as guidance for the gain of gestational weight(13).
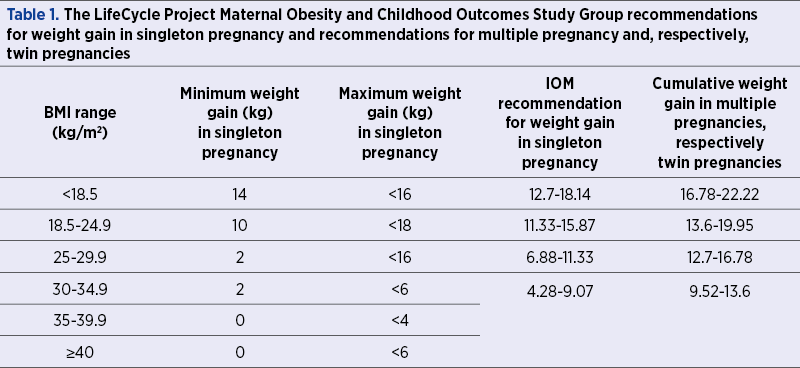
The recommendations for weight gain in singleton pregnancy of the LifeCycle Project Maternal Obesity and Childhood Outcomes Study Group and those of the Institute of Medicine (which are the same with the recommendations of Centers for Disease Control and Prevention), along with the recommendations for multiple pregnancy, respectively twin pregnancy, are summarized in Table 1(11,14-16).
The recommended caloric intake during pregnancy is the normal one for the first trimester, on the consideration that the weekly weight gain should be kept to a minimum. In the second and third trimesters, there is a recommendation for an increase of 300 calories in order to achieve a weekly weight gain of 0.45 kg, but in obese and overweight females the increase should be smaller because the weekly weight gain target is of only 0.23 kg(17). The risk in gestational weight gain above the IOM target is reflected on the pregnancy outcome and on the postpartum outcome. Regarding the pregnancy outcome, the cited risks are: large-for-gestational-age infants, macrosomia, need for caesarean delivery, pregnancy-related high blood pressure (preeclampsia or gestational hypertension) and gestational diabetes(18-20). Regarding the postpartum outcome, for the offspring there is an increased risk of overweight or obesity and of neonatal adiposity, while for the mother, the risk is of postpartum weight retention(21,22). Moreover, in case of macrosomia (a birth weight of more than 4000 grams), there is a higher risk for neonatal brachial plexus injury, clavicle fracture, shoulder dystocia or even caesarean delivery in case of birth weight higher than 5000 grams or, respectively, 4500 grams in case of patients with GDM(23). It is important to emphasize also that the risk of SGA is present in underweight pre-pregnant women, being also described in obese pre-pregnant females(24).
For gestational weight gain below the IOM target, there are also pregnancy and postpartum outcomes. The pregnancy outcomes are represented by the small birth weight and an increased risk for preterm delivery and SGA(18). The postpartum outcomes are SGA and a risk for not retaining weight postpartum, and the SGA risk is the one that should raise awareness(25). The approach for women with weight gain below IOM targets is to evaluate the eating behaviour and the other possible etiologies in order to determine any risks for the fetal growth. The most frequent risk factors are smoking, medical conditions, food insecurity, eating disorders, along with nausea and vomiting from hyperemesis gravidarum. Regarding the management, there may be useful to include the approach of medical or psychiatric conditions, the improvement of the access to healthy foods and changes in diet and concerning the amount of physical activity(26).
In case of pregnant women with a weight gain above the IOM targets, the underweight ones prevent the risk for SGA, but there should be assessed their eating habits and other potential comorbidities, such as oedema (eclampsia, nephropathy or heart failure), maternal adiposity increment or decreased caloric consumption. Those cases need a particular management that should include recommendations regarding diet, exercise(27) and metformin in case of GDM(28).
GDM is defined as glucose intolerance with the first recognition or onset during pregnancy and has an increasing prevalence, including in pregnant females. GDM and other perinatal outcomes, such as preeclampsia, gestational hypertension, macrosomia, preterm birth and caesarean delivery, are highly associated with overweight or obesity prior to pregnancy(29).
Metformin is a drug commonly used in GDM and in type 2 diabetes mellitus, with proven passage from mother to fetus, that has potential benefits both for the newborn and mother, and it decreases the risk of weight gain during pregnancy, of maternal or neonatal hypoglycemia, of gestational hypertension, of admission in the intensive care unit, of low gestational age, large for gestational age and, respectively, it doesn’t increase the risk for fetal and maternal outcomes, such as birth injury, congenital anomaly, preterm birth, the need for caesarean delivery, shoulder dystocia, an Apgar score below 7 at 5 minutes, maternal glycaemic control and preeclampsia as compared to insulin(30).
Conclusions
Weight gain during pregnancy and maternal pre-pregnancy BMI are two important parameters that can prevent intrapartum and postnatal severe complications. A correct management for a pregnancy should assess very carefully the nutritional status, BMI, the medical conditions, eating disorders and the weight gain during pregnancy. There were developed guidelines for the optimal weight gain and for the management of the cases with excessive weight gain, including nutritional measures, physical activity and even drug administration, especially in patients with GDM.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Torloni MR, Betrán AP, Horta BL, et al. Prepregnancy BMI and the risk of gestational diabetes: a systematic review of the literature with meta-analysis. Obes Rev. 2009 Mar;10(2):194-203.
- Naeye L. Maternal body weight and pregnancy outcome. The American Journal of Clinical Nutrition. 1990;52(2):273-279.
- Santos S, Voerman E, Amiano P, et al. Impact of maternal body mass index and gestational weight gain on pregnancy complications: an individual participant data meta-analysis of European, North American and Australian cohorts. BJOG. 2019;126(8):984-995.
- Nowak M, Kalwa M, Oleksy P, Marszalek K, Radon-Pokracka M, Huras H. The relationship between pre-pregnancy BMI, gestational weight gain and neonatal birth weight: a retrospective cohort study. Ginekologia Polska. 2019;90(1):50–54
- Rosenbluh O, Walfisch A. Birth defects associated with obesity. Clin Exp Obstet Gynecol. 2021;48(3):472-477.
- Alavi N, Haley S, Chow K, McDonald SD. Comparison of national gestational weight gain guidelines and energy intake recommendations. Obes Rev. 2013;14(1):68-85.
- Wang M, Freaney P, Perak A, et al. Trends in Prepregnancy Obesity and Association with Adverse Pregnancy Outcomes in the United States, 2013 to 2018. JAHA. 2021;10:e020717.
- Donofry S, Emery R, Kolko Conlon RP, et al. Documenting the course of loss of control over eating prior to, during and after pregnancy among women with pre-pregnancy overweight and obesity. Int J Eat Disorders. 2021;54(4):633-638.
- Stevens D, Neelon B, Roberts J, et al. Mediation of the association between maternal pre-pregnancy overweight/obesity and childhood overweight/obesity by birth anthropometry. Journal of Developmental Origins of Health and Disease. 2021;12(1):71-78.
- Kominiarek MA, Peaceman AM. Gestational weight gain. Am J Obstet Gynecol. 2017 Dec;217(6):642-651.
- Luke B. Improving multiple pregnancy outcomes with nutritional interventions. Clin Obstet Gynecol. 2004;47(1):146-62.
- Klein L. Nutritional recommendations for multiple pregnancy. Journal of the American Dietetic Association. 2005;105(7):1050-2.
- Institute of Medicine, Food and Nutrition Board, Committee on Nutritional Status During Pregnancy, part I: Nutritional Status and Weight Gain. National Academy Press, Washington, DC, 2000.
- LifeCycle Project-Maternal Obesity and Childhood Outcomes Study Group, Voerman E, Santos S, et al. Association of Gestational Weight Gain with Adverse Maternal and Infant Outcomes. JAMA. 2019 May 7;321(17):1702-1715.
- Committee on Obstetric Practice. ACOG Committee Opinion on Weight Gain During Pregnancy. ACOG. 2013:548, reaffirmed 2020:548. Available at: https://www.acog.org/clinical/clinical-guidance/committee-opinion/articles/2013/01/weight-gain-during-pregnancy (accessed in 24.10.2021).
- Center for Disease Control and Prevention. Weight Gain During Pregnancy. Available at: https://www.cdc.gov/reproductivehealth/maternalinfanthealth/pregnancy-weight-gain.htm (accessed in 24.10.2021).
- Weight Gain During Pregnancy: Reexamining the Guidelines. Institute of Medicine (US) and National Research Council (US) Committee to Reexamine IOM Pregnancy Weight Guidelines, Rasmussen KM, Yaktine AL (Eds), National Academies Press (US), The National Academies Collection: Reports funded by National Institutes of Health, Washington (DC) 2009.
- Goldstein RF, Abell SK, Ranasinha S, et al. Association of Gestational Weight Gain with Maternal and Infant Outcomes: A Systematic Review and Meta-analysis. JAMA. 2017 Jun 6;317(21):2207-2225.
- Hedderson MM, Gunderson EP, Ferrara A. Gestational weight gain and risk of gestational diabetes mellitus. Obstet Gynecol. 2010 Mar;115(3):597-604. Erratum in: Obstet Gynecol. 2010 May;115(5):1092.
- Kominiarek MA, Saade G, Mele L, et al. Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Maternal-Fetal Medicine Units (MFMU) Network. Association Between Gestational Weight Gain and Perinatal Outcomes. Obstet Gynecol. 2018 Oct;132(4):875-881.
- Nehring I, Lehmann S, von Kries R. Gestational weight gain in accordance to the IOM/NRC criteria and the risk for childhood overweight: a meta-analysis. Pediatr Obes. 2013 Jun;8(3):218-24.
- Starling AP, Brinton JT, Glueck DH, et al. Associations of maternal BMI and gestational weight gain with neonatal adiposity in the Healthy Start study. Am J Clin Nutr. 2015 Feb;101(2):302-9.
- Nguyen M, Ouzounian J. Evaluation and Management of Fetal Macrosomia. Obstet Gynecol. 2021;48(2):387-399.
- Chen YH, Li L, Chen W, et al. Pre-pregnancy underweight and obesity are positively associated with small-for-gestational-age infants in a Chinese population. Sci Rep. 2019:9:15544.
- Mannan M, Doi SA, Mamun AA. Association between weight gain during pregnancy and postpartum weight retention and obesity: a bias-adjusted meta-analysis. Nutr Rev. 2013 Jun;71(6):343-52.
- Akgun N, Keskin HL, Ustuner I, Pekcan G, Avsar AF. Factors affecting pregnancy weight gain and relationships with maternal/fetal outcomes in Turkey. Saudi Med J. 2017 May;38(5):503-508.
- Renault KM, Nørgaard K, Nilas L, et al. The Treatment of Obese Pregnant Women (TOP) study: a randomized controlled trial of the effect of physical activity intervention assessed by pedometer with or without dietary intervention in obese pregnant women. Am J Obstet Gynecol. 2014 Feb;210(2):134.e1-9.
- Balani J, Hyer SL, Rodin DA, Shehata H. Pregnancy outcomes in women with gestational diabetes treated with metformin or insulin: a case-control study. Diabet Med. 2009 Aug;26(8):798-802.
- Teshome AA, Li Q, Garoma W, et al. Gestational diabetes mellitus, pre-pregnancy body mass index and gestational weight gain predicts fetal growth and neonatal outcomes. Clinical Nutrition ESPEN. 2021;42:307-312.
- He K, Guo Q, Ge J, Li J, Li C, Jing Z. The efficacy and safety of metformin alone or as an add-on therapy to insulin in pregnancy with GDM or T2DM: A systematic review and meta- analysis of 21 randomized controlled trials. J Clin Pharm Ther. 2021 Aug 7. doi: 10.1111/jcpt.13503. Epub ahead of print.
Articole din ediţiile anterioare
Actualităţi în managementul afecţiunilor tiroidiene la femei în perioada sarcinii
Afecţiunile tiroidiene sunt printre cel mai frecvent întâlnite patologii la femeile aflate la vârsta fertilă. Homeostazia tiroidiană este important...
Rolul 25 hidroxivitaminei D (25(OH)D) în travaliul prematur
Travaliul prematur este cea mai importantă cauză de deces în cazul copiilor sub 5 ani, la nivel mondial. Etiologia travaliului prematur este adesea...
Impactul endometriozei asupra sarcinii
Endometrioza şi adenomioza sunt patologii care afectează frecvent femeile de vârstă reproductivă. Obţinerea şi menţinerea unei sarcini se fac de mu...
Cât de des recomandaţi suplimentarea cu vitamină D în sarcină? Ce trebuie să ştie obstetricienii despre vitamina D şi sarcină
Vitamina D (calciferolul), măsurată prin 1,25-dihidroxivitamina D în serul matern, este importantă pentru dezvoltarea unităţii fetoplacentare. Celu...