Introduction. Customizing surgical radicality is a crucial element in contemporary cervical cancer surgery. Currently, anatomic structures are characterized based on many anatomic interpretations. In order to suggest its general applicability, this study proposes to standardize the definitions and anatomic landmarks of the various types of radical hysterectomy. Methodology. This paper is a literature review in which we selected relevant articles from PubMed database, starting with the year of publication 2010 until 2023, using the following keywords: “cervical cancer”, “radical hysterectomy”, “classification of radical hysterectomy”, “anatomic landmarks”, “tailoring surgery”, “nerve-sparing procedure”. Results. The lateral extent of resection serves as the basis for the updated Querleu-Morrow classification. The three-dimensional anatomic template used to define the resection limits is based on the precise anatomy of the paracolpium and parametrium. The main anatomic landmarks and avascular spaces of the pelvis serve as the basis for the oncologic procedures, including nerve-sparing techniques. There are four different types of radical hysterectomy, each type described with three-dimensional landmarks. There are three main objectives that never change: excising the central tumor with clear margins, removal of any possible lymph dissemination sites, and the control of metastatic disease. Conclusions. It is essential to base research evaluating radicality in the surgical treatment of cervical cancer on precise and generally accepted nomenclature and descriptions. Since the degree of parametrium excision affects late morbidity, particularly bladder and rectal dysfunctions, the thorough explanation and understanding of the many forms of radical hysterectomy are crucial.
Anatomic landmarks in radical hysterectomy
Repere anatomice în histerectomia radicală
First published: 30 iunie 2023
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/ObsGin.71.2.2023.8870
Abstract
Rezumat
Obiective. Personalizarea limitelor excizionale reprezintă un element de bază în chirurgia contemporană a cancerului de col uterin. În prezent, structurile anatomice sunt caracterizate pe baza mai multor interpretări anatomice. Pentru a sugera aplicabilitatea sa generală, acest studiu a avut drept scop standardizarea definiţiilor şi reperelor anatomice ale diferitelor tipuri de histerectomie radicală. Materiale şi metodă. Această lucrare reprezintă o revizuire a literaturii de specialitate, în cadrul căreia am selectat cele mai relevante articole din baza de date PubMed, începând cu anul publicării 2010 până în 2023, folosind următoarele cuvinte-cheie: cancer de col uterin, histerectomie radicală, parametru, paracolpos, clasificarea histerectomiei radicale, repere anatomice, chirurgie personalizată, procedură de prezervare a nervilor. Rezultate. Modelul anatomic tridimensional utilizat pentru definirea limitelor de rezecţie se bazează pe anatomia precisă a paracolposului şi a parametrului şi reprezintă baza clasificării actualizate Querleu-Morrow. Principalele repere anatomice şi spaţiile avasculare ale pelvisului stau la baza oricărei proceduri cu viză oncologică. Există patru tipuri diferite de histerectomie radicală, fiecare tip descris cu repere anatomice. Există trei obiective principale general valabile: excizia tumorii centrale cu margini de siguranţă, bilanţul diseminării ganglionare şi controlul bolii metastatice. Concluzii. Diferite interpretări, nomenclaturi şi artefacte de disecţie din literatură fac dificilă standardizarea reperelor anatomice. Deoarece complicaţiile postoperatorii – în special disfuncţiile vezicii urinare şi ale rectului – sunt direct corelate cu limitele excizionale ale parametrelor, o explicaţie amănunţită şi o înţelegere a numeroaselor tipuri de histerectomie radicală sunt esenţiale.
Introduction
Cervical cancer provides one of the few examples of controversial treatment management throughout medical history. Radical hysterectomy was first described and performed by Clark and Reis in 1895(1). Later on, Wertheim and his mentor Shauta established the surgical principles and details of the operation, followed by an intense disagreement between them regarding whether cervical cancer should be treated with abdominal or vaginal approach(1). In the early 20th century, radical hysterectomy was abandoned as treatment option by many gynecologists due to radiotherapy (RT) development; however, in 1950, Meigs modified and repopularized radical hysterectomy(1). It is very important to mention the contribution brought by Okabayashi to the prevention of bladder dysfunction by preserving the pelvic splanchnic nerves and bladder branch of the inferior hypogastric plexus(1).
Radical hysterectomy has undergone multiple changes over time, thanks to the efforts of many great surgeons and scientist, being still continuously improved by current pioneers and contributors. Nowadays, cervical cancer surgery principles are based on tailoring procedure radicality so that the optimal technique is personalized according to the tumor size and infiltration in the vaginal vault.
Different interpretations, nomenclatures and artificial dissection artifacts from literature make it difficult to standardize anatomic landmarks. The term “radical” is currently used to describe different techniques, each one with another degree of parametrial extension. The most popularized and accepted classifications of radical hysterectomy are made by Piver, dating since 1974, and Querleu-Morrow, published in 2008, both based on lateral extent of resection(2,3). Still, there are many unmet needs of standardization regarding the adjustment of radicality according to different stages of International Federation of Gynecology and Obstetrics (FIGO) classification, taking into account especially the three-dimensional patterns of tumor spreading(4).
Materials and method
The aim of this paper was to analyze data from literature in order to standardize the anatomic landmarks in radical hysterectomy. This paper is a literature review in which we selected relevant articles from PubMed database, starting with the year of publication 2010 until 2023, using the following keywords: “cervical cancer”, “parametrium”, “paracolpos”, “radical hysterectomy”, “classification of radical hysterectomy”, “anatomic landmarks”, “tailoring surgery”, “nerve-sparing procedure”. The fertility sparing surgery data were not included.
Results
Pelvic avascular spaces and anatomic landmarks
Many authors advocate the need for a generally valid and accepted anatomic nomenclature. The current trend is in favor of using ventral-dorsal, caudal-cranial and medial-lateral terms, rather than anterior-posterior, deep-superficial and internal-external, that are more likely to be confusing(4).
The exact anatomy of the parametrium and paracolpium forms the basis of the three-dimensional anatomic template for defining the resection limits(4). It also takes into account the ureter’s stable anatomic landmark status, which divides the paracervix into two sections: the parametrium (above, cranial and craniomedial from the ureter) and the paracolpium (beneath, caudal and caudolateral from the ureter)(4).
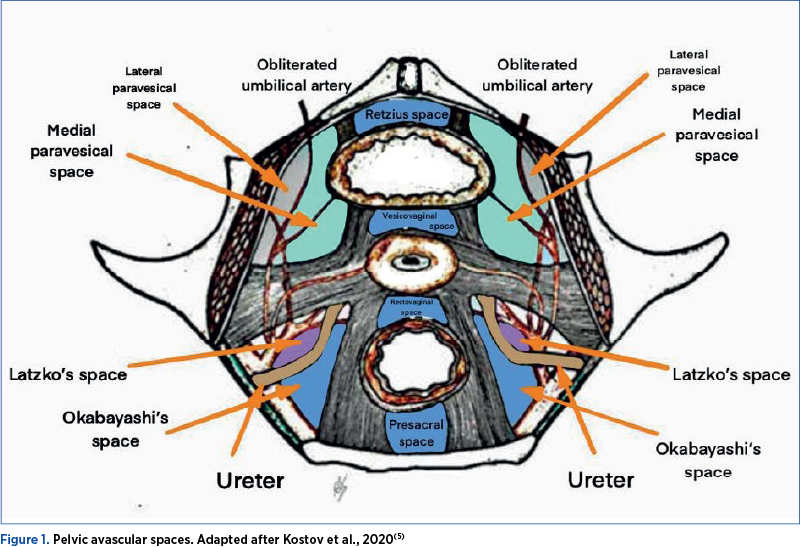
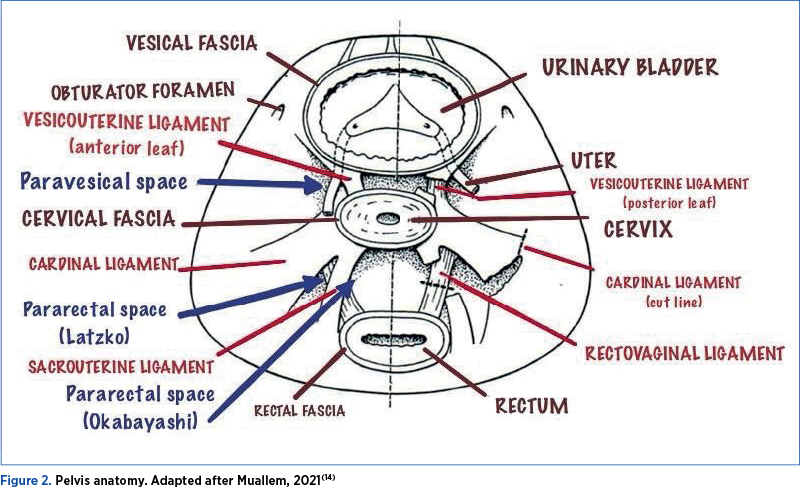
The pelvic avascular spaces are divided in lateral and median spaces (Figure 1). Lateral spaces consist in paravesical space (divided by the umbilical artery into medial paravesical space and lateral paravesical space), pararectal space (divided by the ureter into lateral pararectal space (Latzko space) and medial pararectal space (Okabayashi space), and the fourth space (Yabuki space)(5). Median spaces are represented by retropubic (Retzius), vesico-cervical/vesico-vaginal, rectovaginal and presacral spaces(5).
Paravesical space boundaries are: superior pubic ramus and arcuate line of ilium (ventral), cardinal ligament including parametrium and paracervix (dorsal), caudal portion of vesico-uterine ligament and bladder (medial), obturator internus muscle and external iliac vessels (lateral), anterior leaf of cardinal ligament (cranial), iliococcygeus muscle and pubocervical fascia (caudal)(6). Paravesical space contains the umbilical artery, the superior vesical artery, the lymphatic tissue and the obturator neurovascular bundle(6). It is important to mention that, during the dissection of this space, the surgeon has to be aware of the presence of corona mortis (vascular anastomosis between the external iliac and obturator vessels)(7).
Pararectal space borders consists in: cardinal ligament including the parametrium, over the ureter part, and paracervix (ventral), presacral fascia and sacrum (dorsal), internal iliac artery (lateral), rectum (medial), posterior leaf of the cardinal ligament (cranial) and levator ani muscle (caudal)(8). The ureter divides the pararectal space into the Okabayashi space, located medially, and the Latzko space, located laterally (Figures 1 and 2)(8).
The borders of Okabayashi space are levator ani (dorsal), uterine artery (superior), posterior leaf of the broad ligament (cranial and ventral), rectum (medial) and ureter (lateral)(8). The lateral ligament of the rectum is formed by middle rectal artery and vein, and it serves as important landmark in nerve-sparing procedures(8). The lateral ligament of the rectum transverse the pelvic plexus and is located near the pelvic splanchnic nerves(8).
Latzko’s space is formed by the cardinal ligament (ventral), parasacral fascia (dorsal), internal iliac artery (lateral) and ureter (medial)(8). When pelvic lymphadenectomy or inferior hypogastric plexus (pelvic plexus) access is needed, Latzko’s space is a helpful anatomic landmark(8). The uterine artery that arises from the internal iliac artery is visible following the dissection of Latzko’s space(8).
The fourth space was first defined by Yoshihiko Yabuki in 2000, and it lies between the cranial portion of the vesicouterine ligament and the ureter(9). It is important especially in nerve sparing procedures due to splanchnic nerves pathways(9).
The Retzius space borders are: pubic symphysis (ventral), bladder and parietal peritoneum (dorsal), transversals fascia (cranial), pubocervical fascia, bladder neck and urethra (caudal) and arcus tendinous fasciae pelvis (lateral)(10). It contains a large plexus of vein which drains into the internal iliac vein. It can be identified within the paravaginal tissue, known as Santorini plexus(10).
The vesicovaginal space, also called anterior cul-de-sac, is surrounded by the bladder trigone (ventral), pubocervical fascia and vagina (dorsal) and by the cranial part of the vesicouterine ligament (lateral)(11). Vaginal cuff resection requires vesicovaginal space dissection(11). To prevent bleeding and the loss of the correct dissection plane, it is crucial to remain above the pubovesical fascia. It is important to acknowledge the dictum “fat belongs to the bladder”(11). The components of cranial vesicouterine ligament are: uterine artery, superficial uterine vein, ureter branch of the uterine artery, superior vesical vein that drains into the superficial uterine vein and cervicovesical vessels(11). The cranial and caudal sections of the vesicouterine ligament are separated by the ureter(11).
The rectovaginal space is formed by posterior vaginal wall (ventral), anterior rectal wall (dorsal), uterosacral ligaments and rectovaginal ligament (lateral), peritoneal reflection of the Douglas pouch (cranial) and levator ani muscle (caudal)(12). The dissection of this place should be made between the two layers of Denonvilliers’ fascia, in order to avoid vaginal bleeding or rectal injuries(12). There are three vascular structures which the surgeon should be aware of: middle rectal artery and vein, vaginal veins, and presacral veins(12).
The presacral space limits are mesorectal fascia and rectum (ventral), sacral promontory and longitudinal anterior vertebral ligament (dorsal), common iliac and ureter (lateral), peritoneal reflection of the rectosigmoid (cranial) and pelvic floor (caudal)(13). The Waldeyer’s fascia separates this region into inferior and superior retrorectal spaces(13). The anatomic structures which require increased attention are common iliac vessels, inferior mesenteric artery, middle sacral vessels, and ureters(13).
Classification of radical hysterectomy
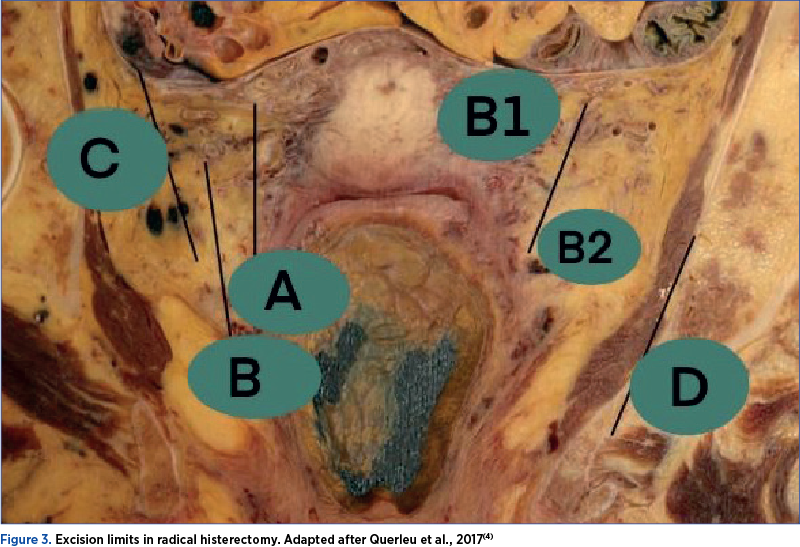
The updated Querleu-Morrow classification system (Figure 3) emphasizes the role of lateral extent of resection using clear anatomic landmarks, such as the cross between ureter and uterine artery, ureter and paracervix and the vascular plane of internal iliac vessels(4). The following description of each type include Cibula 3-D concept(4,11).
Type A procedure includes minimum resection of the paracervix(15). The location of ureter must be determined under direct visualization(15). The uterine artery can be transected at the cross with the ureter or at the origin(15). In order to reach and open the lateral vaginal fornix, the paracervix is excised caudally parallel to the cervix and halfway between the ureter and the cervix(15). There is no paracolpos or vaginal portion of the paracervix removed in this procedure(15). The amount of vaginal excision is usually less than 10 mm(15). This hysterectomy type excludes the possibility of resecting the dorsal parametria, as well as the ventral and lateral parametria, thus the hypogastric plexus is completely intact(15).
Type B technique is focused on transection of paracervix at the ureter level(4). A horizontal excision of around 1 to 1.5 cm of the lateral parametria is possible, since the resection boundary is at the medial aspect of the ureteral bed(4). The ureteral artery (an important anatomic landmark), which arises from the uterine artery, is usually easily identified and can be preserved(15). In the dorsal parametrium, removal margin is located at the level of the vaginal cuff excision, but due to branches of the hypogastric plexus, it cannot extend beyond the course of the ureter(4). Type B subdivides in type B1 procedure (as described above) and type B2 surgery (identical to B1, but combined with pelvic lymphadenectomy)(4). The paracervical lymphadenectomy includes the removal of the lymph nodes cranial to the sciatic nerve and caudal to the obturator nerve(4,15). The landmark for safely entering the retroperitoneal space is the external iliac artery(4).
Type C procedure consists in transection of paracervix at the junction with the internal iliac vascular system(4). It is tailored for FIGO stage IB1-2A or early 2B cervical malignancies(4). The medial side of the internal iliac vein and artery is known as the lateral limit for excision(4). The ureter is fully lateralized and mobilized(4). The vaginal cuff’s excision length is modified based on the tumor’s vaginal extension(4). The rectovaginal and rectouterine ligaments are transected at the rectum(4). At the bladder, the ventral parametrium ligament is transected(4). The resection is performed on the vesicouterine and vesicovaginal ligaments(4). In order to perform type C1 procedure, the dorsal parametrium must be divided into two sections: the lateral laminar structure, also known as the mesoureter, which houses the hypogastric plexus, and the medial component, which includes the rectouterine and rectovaginal ligaments(4,15). In C1 type, the ureter from the ventral parametrium only needs to be partially dissected(4). The ureter is fully dissected in the C2 type, from the ventral parametria to the wall of the bladder(4,15). The medial side of the internal iliac vein and artery forms the same lateral border for both types(4).
Type C1. Regarding longitudinal resection margins in the lateral parametrium, the vaginal vein forms the deep parametrial resection boundary, protecting the caudal portion of the lateral parametrium that contains the splanchnic nerves(4). When the ureter is partially detached from the ventral parametrium, 1 to 2 centimeters of the ventral parametrium can be excised(4). The resection line is formed by the bladder branches of the hypogastric plexus, which are located below the ureter’s course and act as longitudinal boundaries(4). In the dorsal parametrium, the hypogastric nerves from the rectouterine and rectovaginal ligaments must be sagittally dissected(4). The tangential plane of the vaginal cuff excision forms the caudal limit on the rectouterine and rectovaginal ligaments, while the main branches of the hypogastric plexus must be preserved on the lateral part(4,15).
Type C2. In the lateral parametrium, the resection line extends caudally up to the pelvic floor (sacral bone), paralleling the medial aspect of the internal iliac and pudendal arteries(4). The splanchnic nerves in the caudal region are sacrificed in order to fully unite the pararectal and paravesical spaces(4). In order to fully remove the ventral parametrium up to the wall of the bladder, complete dissection of the ureter is necessary; the medial and lateral leafs of the ventral parametrium are resected equally(4). In longitudinal aspect, vaginal resection and the paracolpium’s level combine to generate the resection line(4). The ventral parametrium’s cranial and caudal (below the ureter) portions are eliminated(4). Identification of the hypogastric plexus bladder branches is not necessary, since they are sacrificed. In type C2 radical hysterectomy, the excision of dorsal parametrium is mandatory(4).
Type D. Similar to type C2, ureteral dissection and excision of both dorsal and ventral parametrium occur(4,15). This procedure requires the ligation and excision of the internal iliac artery and vein, as well as its branches, which include the internal pudendal, gluteal and obturator veins(4,15). The piriformis muscle, obturator internal muscle, and lumbosacral nerve plexus comprise the lateral resection line(4).
Radicality and tailoring procedures
Tumors classified as FIGO IB2, IB3 (voluminous but without macroscopic infiltration in the vaginal vault or obvious infiltration in parametrium/paracolpium) require a tailored technique. To close the vaginal cuff beneath the tumor (caudal) and prevent tumor cells from spreading and contaminating the abdominal cavity, these tumors demand the removal of a longer vaginal vault(14). Tumors with ventral localization in these circumstances require the removal of a longer vaginal cuff compared to tumors of the same size and stage but with dorsal localization because of the curved and anteflexed nature of the upper vagina(14). As well, tumors classified as FIGO IIA1, IIA2 (tumors that infiltrate the vaginal wall, with no obvious infiltration in parametrium/paracolpium) necessitate a different approach in terms of radicality. In order to perform a tumor excision within oncological safety limits, these tumors require the resection of a longer vaginal vault with paracolpium together with the blood vessels and lymph nodes from the upper third to the middle of the vagina(14). Both situations are operable with possible nerve-sparing technique in order to reduce to the minimum the postoperative complications(14).
Discussion
The anatomic landmarks in radical hysterectomy represent the basis in terms of oncological precision, as well as the safety of the entire medical act. Thus, it is mandatory that every surgeon recognizes these structures and their anatomic variations. The anatomic landmarks for oncologic pelvic procedures, including nerve-sparing techniques, are: the four spaces (medial paravesical, Okabayashi, Latzko, and Yabuki spaces) and four structures (the ureter, lateral ligament of the rectum, middle rectal artery, and the deep uterine vein)(9).
Currently, the various forms of radical hysterectomy are classified and named using different nomenclature. Setting standards for performing genuine multicenter surgical trials and sharing data are all severely hampered by the lack of a universally acknowledged classification system.
Conclusions
Since the degree of parametrium excision affects late morbidity, particularly bladder and rectal dysfunctions, the thorough explanation and understanding of the many forms of radical hysterectomy are crucial.
Research assessing radicality in the surgical treatment of cervical cancer requires to be established on accurate, widely recognized descriptions and nomenclature. It is obvious that a globally recognized classification system for radical hysterectomy with clear anatomic landmarks is required, one that research teams, individual surgeons, national and international societies will all be able to use.
Conflict of interest: none declared
Financial support: none declared
This work is permanently accessible online free of charge and published under the CC-BY.

Bibliografie
- Dursun P, Gultekin M, Ayhan A. The history of radical hysterectomy. J Low Genit Tract Dis. 2011;15(3):235-45.
- Piver MS, Rutledge F, Smith JP. Five classes of extended hysterectomy for women with cervical cancer. Obstet Gynecol. 1974;44(2):265-272.
- Querleu D, Morrow CP. Classification of radical hysterectomy. Lancet Oncol. 2008;9(3):297-303.
- Querleu D, Cibula D, Abu-Rustum NR. 2017 Update on the Querleu-Morrow Classification of Radical Hysterectomy. Ann Surg Oncol. 2017;24(11):3406-3412.
- Kostov S, Slavchev S, Dzhenkov D, Mitev D, Yordanov A. Avascular Spaces of the Female Pelvis-Clinical Applications in Obstetrics and Gynecology.
- J Clin Med. 2020;9(5):1460.
- Selçuk İ, Ersak B, Tatar İ, Güngör T, Huri E. Basic clinical retroperitoneal anatomy for pelvic surgeons. Turk J Obstet Gynecol. 2018;15(4):259-269.
- Sanna B, Henry BM, Vikse J, et al. The prevalence and morphology of the corona mortis (Crown of death): A meta-analysis with implications in abdominal wall and pelvic surgery. Injury. 2018;49(2):302-308.
- Gaspar H, Arencibia Sanchez O, Jordi Ponce J. Textbook of Gynecological Oncology – Surgical Anatomy. In: Pelvic Gynecologic Oncology, 3rd ed., 2016, ESGO Academy: Brussels, Belgium.
- Lemos NL, Ribeiro R, Fernandes GL, Abrão M, Moretti-Marques R. Nerve-sparing routes in radical pelvic surgery. In: Minimally Invasive Gynecology; Springer: Berlin/Heidelberg, Germany, 2018; pp. 61–75.
- Muavha DA, Ras L, Jeffery S. Laparoscopic surgical anatomy for pelvic floor surgery. Best Pract Res Clin Obstet Gynaecol. 2019;54:89-102.
- Ansari MM. Retzius Space: Not A Single Anatomical Entity: New Insights Simplified and Illustrated in A Laparoscopic Study during TEPP Hernioplasty for Inguinal Hernia. Ann Int Med Den Res. 2018;4:SG63–SG73.
- Fujii S, Sekiyama K. Precise Neurovascular Anatomy for Radical Hysterectomy. Springer Nature Singapore PTE LTD, 2020.
- Patel N, Maturen KE, Kaza RK, Gandikota G, Al-Hawary MM, Wasnik AP. Imaging of presacral masses – a multidisciplinary approach. Br J Radiol. 2016;89(1061):20150698.
- Muallem MZ. A New Anatomic and Staging-Oriented Classification of Radical Hysterectomy. Cancers (Basel). 2021;13(13):3326.
- Cibula D, Abu-Rustum NR, Benedetti-Panici P, Köhler C, Raspagliesi F, Querleu D, Morrow CP. New classification system of radical hysterectomy: emphasis on a three-dimensional anatomic template for parametrial resection. Gynecol Oncol. 2011;122(2):264-8.