Lung cancer is the second most frequent malignancy and the most common cause of cancer-related death in both men and women. In this article, we will present the case of a non-smoker patient, with left laterocervical lymph nodes at diagnostic, with unknown primary origin. After PET-CT and IHC immunohistochemistry, he is confirmed with lung tumor (adenocarcinoma), pleural, bone, thyroid and salivary gland metastases, the last two being surgically removed. The patient received chemotherapy for 4 cycles with platinum doublet, and afterwards switched to maintenance with pemetrexed. Due to very good response to treatment, he is downstaged and receives curative RT for lung lesion. Currently, he continues maintenance with Alimta, the disease being in complete remission.
Cancer pulmonar non-microcelular cu metastaze tiroidiene și de parotidă la diagnostic și remisiune completă după tratament. Prezentare
NSCLC with thyroid and parotid metastases at diagnostic, with complete remission after treatment. Case presentation
First published: 25 aprilie 2017
Editorial Group: MEDICHUB MEDIA
Abstract
Rezumat
Cancerul pulmonar este a doua cea mai frecventă neoplazie şi prima cauză de deces asociată neoplaziilor atât la bărbaţi, cât şi la femei. În acest articol, vom prezenta cazul unui pacient nefumător, cu adenopatie laterocervicală stângă la momentul diagnosticului, fără punct de plecare precizat. După efectuarea PET-CT şi a testelor de imunohistochimie, este confirmat cu tumoră pulmonară (adenocarcinom) şi metastaze pleurale, osoase, tiroidiene şi de glandă salivară, ultimele două fiind extrase chirurgical. Pacientul a primit chimioterapie 4 serii cu dublet de platină şi ulterior a fost trecut pe mentenanţă cu pemetrexed. Datorită răspunsului foarte bun la tratament, este restadializat şi primeşte radioterapie cu intenţie curativă pentru tumoră pulmonară. La momentul actual, continuă tratamentul cu Alimta, boala fiind în remisiune completă.
Case presentation
A male patient, 48-years-old, from urban area, presents at clinic in July 2014.
History: April 2014 - he presents at the hospital for laterocervical lymph nodes, surgically removed.
Risk factors: non-smoker, without professional exposure, alcohol occasionally.
Comorbidities: surgery for repair of mitral valve after endocarditis (2002) and surgery for femural head avascular necrosis (2011).
Physical examination: scar in cervical region (after surgery), pulmonary - dullness and diminished murmur in lower left lung, BP: 140/70 mmHg, HR: 72/min, left vocal cord paralysis.
Laboratory findings:
Blood tests were in normal range, except LDH: 509 U/L (170-480U/L).
Computer tomography (CT) of cervical area, thorax and abdomen, in April 2014, found left pulmonary lesion without specificity, thin, 8 mm fluid accumulation in left costodiafragmatic sinus.
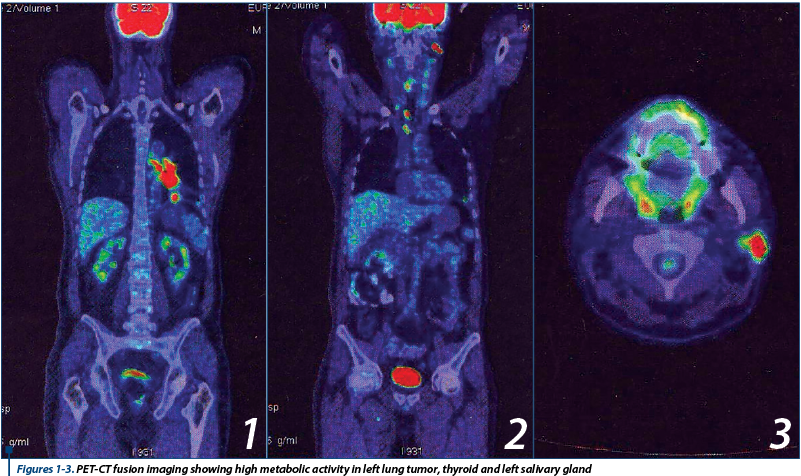
Because of the presence of laterocervical tumoral lymph nodes with unknown primary origin, a PET-CT is ordered in July 2014, that showed high metabolic activity in left lower lung tumor 38/40 mm (Figure 1), in multiple cervical, mediastinal and hilar lymph nodes, as well as in lesions at thyroid (Figure 2) and parotid level (Figure 3). PET-CT also showed two infracentimetric osteocondensant lesions on ribs VII and VIII (interpreted as post traumatic). Suspect bone lesion on right iliac wing and sacroiliac articulation. Bilateral osteolytic lesions on hip head, accentuated on the right one (suggestive for aseptic osteonecrosis - known from patient history).
Bronhoscopy (and cytology) only offered indications about category of tumor (SCLC or NSCLC - in our case) without being able to orient us regarding exact histology (adenocarcinoma, squamos, or large cell).
With results from PET-CT and bronhoscopy, and needing to asses exact tumor type, most accessible lesions are removed (June 2014 - left thyroid lobe, left parotid) and histopathology and IHC examination requested. They proved to be metastasis of lung adenocarcinoma.
After suspicion of bone metastasis on PET CT, scintigraphy is recommended to bring more details regarding bone lesions, which shows right hip head hyperfixation (aseptic necrosis in revascularization state), suspicious hyperfixating lesion on right sacroiliac articulation, which needs monitoring. No intervention is taken regarding the bone, with close monitoring of current lesions.
Genetic tests: EGFR mutations exons 18, 19, 20, 21 - not present.
Diagnostic
Clinical suspicion of lung neoplasm, sustained by imagistic studies (PET-CT), and confirmed by histopathology and immunohistochemistry results.
Final diagnostic: non small cell lung cancer (adenocarcinoma) of left pulmonary lobe with pleural, bone, thyroid and salivary gland metastasis (last two operated), stage IV.
Differential diagnostic: neoplasm of thyroid and neoplasm of salivary gland - which were differentiated by immunohistochemistry.
Treatment
Because of present symptomatology and patient preference to start treatment as soon as possible, we opted for chemotherapy - platinum doublet.
First line: patient was given 4 cycles of cisplatin 75mg/m2 and paclitaxel 175mg/m2 every 21 days.
In this circumstance of good response to treatment (shrinkage of primary tumor, no new lesions, good PS-performance score) and operated metastasis, he is downstaged and directed to radiotherapy consult for potential curative RT for lung + remaining lymph nodes. He has undergone conformational RT on target volume and lymph nodes area, total dose 60 Gy, well supported.
In January 2015, CT - taking into account that the measurable lesions diminished in biggest diameter more then 50% and there are no new lesions, radiologist and clinician concluded that there is a partial response.
After finalizing RT, patient is continued on Alimta maintenance, with good results, that confirm the data from studies that registered the concept of switch maintenance pemetrexed.
Toxicity
After 1 cycle - CINV (chemotherapy induced nausea and vomitting) grade 2 (all toxicities were graded using CTCAE 4.03 - June 14, 2010) - managed by supportive treatment.
After 2 cycles - neutropenia grade 3, required administration of filgrastim and delay of cycle 3 with 2 days ; mucositis grade 2 - symptomatic treatment.
After 4 cycles - sensory peripheral neuropathy grade 2; sent to neurologic consult - symptomatic treatment.
Toxicity - after 7 cycles, grade 3 sensory peripheral neuropathy - referral to neurologic consult; did not require dose reduction/ interruption of treatment.
After 18 cycles, in January 2016 - neurologic symptoms occur - numbness and muscular weakness in right hand (motor neuropathy grade 3) - initially considered a potential clinical sign of brain metastasis.
Neurologic consult shows right radial nerve palsy; it was recommended MRI for cerebral and cervical region. MRI is in normal limits (infirms M1BRA), so electromiography (EMG) is recommended. EMG showed axonal sensitive neuropathy in all limbs, axonal mononeuropathy of radial nerve with important acute denervation (suggestive for lesion of posterior interosseous nerve) and recommends monitoring. Pemetrexed has among rare adverse reaction (<1%) motor neuropathy - this could be the cause of patient’s suffering. If symptomatology worsens, we can take in account discontinuing/stopping this treatment. Current guidelines don’t specify the duration of maintenance treatment and patient has over 6 months with no signs of disease evolution(1). Motor neuropathy is considered of mixed etiology, and treatment with Alimta is continued.
Prognostic
Bearing in mind that initial staging offers the 5-year estimated survival rate (for stage IV under 5%), regardless the very good answer to combined treatment, probability of future disease progression is high.
Close term prognostic, as long as response to treatment is maintained, is good. Medium term prognosis is relatively good. Long term prognosis is a reserved one.
The patient has never smoked and it has been demonstrated that these patients have better prognostic and respond better to chemotherapy. Some data suggest the opportunity to include these patients in a different subgroup(2).
In this case, although EGFR mutation was not detected, and there were numerous initial metastasis sites, response to treatment and survival are very good (present time: 1 year and 8 months).
Evolution
In January 2016, CT TAP - without tumoral lesions in mediastinum, pleural or lung; no enlarged lymph nodes, no lesions suspected for metastasis, right iliac wing - 7 mm lesion suggestive for osteoblastoma (a rare primary neoplasm of bone, categorized as a benign bone tumor that is closely related to osteoid osteoma). Patient will receive 21 days cycles of maintenance chemotherapy until the disease progression or inacceptable toxicity.
Discussions
Maintenance chemotherapy gained an important place in the treatment of NSCLC. Regarding maintenance with pemetrexed (Alimta), there are two important studies that demonstrate the role of this drug in continuous and switch maintenance.
In Paramount study, Pemetrexed continuation maintenance therapy was well tolerated and it was obtained a superior overall survival compared with placebo. This study demonstrated that continuous maintenance is an effective treatment strategy for patients with advanced non squamous non small-cell lung cancer and good performance status that did not progress during pemetrexed-cisplatin induction therapy(3).
The benefit of switch maintenance was demonstrated by JMEN study. Patients who had received four cycles of initial platinum-based chemotherapy and didn’t show progression were randomized 2:1 to active chemotherapy versus placebo every 3 weeks. Both groups received vitamin B12 and folic acid, as is standard for Alimta. The trial was clearly positive for demonstrating a benefit in progression-free survival as well as higher response rates achieved in patients overall who received Alimta after initial platinum-based doublet.
The conclusion of this study was that maintenance therapy with pemetrexed is well tolerated and offers improved progression-free and overall survival compared with placebo in patients with advanced non-small-cell lung cancer(4).
The maintenance with pemetrexed was chosen because EGFR was not present, testing ALK was not performed because at the time of diagnosis, the test was not available in Romania. At this moment, ALK test determination is taken into consideration because now is possible to be performed in Romania (not also ROS1 test) and if it will be positive, treatment with ALK inhibitors is indicated(5,6).
A study by Kalikaki et al., cited by A.C. Grigorescu, demonstrated that “EGFR and K-RAS mutations (which can be found in 8.2% and 22.6% of NSCLC) have a predictive role for response in front-line chemotherapy. These authors stated that EGFR mutation is a prognostic factor for response to chemotherapy and survival and is more frequently found in adenocarcinoma”(7,8).
In our case, despite the negative EGFR mutation status, patient had a good evolution because he was a nonsmoker and the histology was adenocarcinoma.
In the future, if the patient will have progressive disease even if EGFR is not mutated and ALK has not rearrangement, patient can receive erlotinib in the second and third line(9,10). The BR21 study, by Frances AS et al., demonstrated that erlotinib is effective and could prolong survival as second-line treatment for patients with advanced NSCLC without taking into consideration the EGFR mutation status(11).
Immunotherapy could be a therapeutic option taking in consideration the disease evolution and patient performance status, nivolumab (registration trial CheckMate 057) or pembrolizumab (data from trial KEYNOTE-001), latter requiring testing for PD-L1 expression, being at this moment used for treatment of metastatic NSCLC if evolution occur after chemotherapy or target therapy(12,13).
Unfortunately, in the long run, resistance to almost all therapies is developed - including in immunotherapy, as suggested by data(14).
Case particularities
This is a classical case vignette for the switch maintenance with pemetrexed. Other particularity is that this case confirms the good response of adenocarcinoma to pemetrexed and also better response to treatment for nonsmokers.
Our case has demonstrated that association between chemotherapy and radiotherapy is beneficial and in sequential association.
The late, complete remission (1 year and 8 month from diagnostic) is another particularity which should be highlighted.
A particularity is also the atypical metastasis discovered on PET-CT (metastasis of thyroid lobe and salivary gland).
The originality of this case consist also in pseudo bone lesions presented as metastasis on PET-CT/scintigraphy, which proves finally to be of benign origin (osteoblastoma).
We can cite for fine feature rare toxicity to Alimta treatment (right hand motor neuropathy) but its etiology is likely confounding.
Bibliografie
2. Ilka Lopes Santoro, Roberta Pulcheri Ramos, Juliana Franceschini, Sergio Jamnik, and Ana Luisa Godoy Fernandes. Non-small cell lung cancer in never smokers: a clinical entity to be identified, Clinics (Sao Paulo). 2011 Nov; 66(11): 1873–1877.
3. Luis G. Paz-Ares, Filippo de Marinis, Mircea Dediu, Michael Thomas, Jean-Louis Pujol, Paolo Bidoli, Olivier Molinier, Tarini Prasad Sahoo, Eckart Laack. PARAMOUNT: Final Overall Survival Results of the Phase III Study of Maintenance Pemetrexed Versus Placebo Immediately After Induction Treatment With Pemetrexed Plus Cisplatin for Advanced Nonsquamous Non-Small-Cell Lung Cancer. JCO July 8, 2013.
4. Ciuleanu T, Brodowicz T, Zielinski C. Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: a randomised, double-blind, phase 3 study. JMEN Lancet. 2009 Oct 24; 374(9699):1432-40. doi: 10.1016/S0140-6736(09)61497 Epub 2009 Sep 18.
5. K.M. Kerr, L. Bubendorf, M.J. Edelman. ESMO Consensus Guidelines: Pathology and molecular biomarkers for non-small-cell lung cancer. Ann Oncol (2014) 25 (9): 1681-1690. doi: 10.1093/annonc/mdu145
6. Shaw AT, Ou SH, Bang YJ. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N Engl J Med. 2014 Nov 20; 371(21):1963-71. doi: 1056/NEJMoa1406766. Epub 2014 Sep 27.
7. A. C. Grigorescu, C. Bala. Phase II study of erlotinib plus gemcitabine in first-line treatment of poor prognosis, advanced non-small cell lung cancer patients; J BUON 2013; 18(1): 188-194 ISSN: 1107-0625 www.bu-on.org/jbuon.
8. Kalikaki A, Koutsopoulos A, Hatzidaki D. Clinical outcome of patients with non-small cell lung cancer receiving front-line chemotherapy according to EGFR and K-RAS mutation status. Lung Cancer 2010; 69:110-115.
9. Ciuleanu T, Stelmakh L, Cicenas S, Miliauskas S, Grigorescu AC. Efficacy and safety of erlotinib versus chemotherapy in second-line treatment of patients with advanced, non-small-cell lung cancer with poor prognosis (TITAN): a randomised multicentre, open-label, phase 3 study. Lancet Oncol. 2012 Mar; 13(3):300-8. doi: 10.1016/S1470-2045(11)70385-0. Epub 2012 Jan 24.
10. Fiala O, Pesek M, Finek J, Krejci J. Second line treatment in advanced non-small cell lung cancer (NSCLC): comparison of efficacy of erlotinib and chemotherapy. Neoplasma. 2013; 60(2):129-34.
11. Frances AS, Pereira JR, Ciuleanu T et al., Erlotinib in Previously Treated Non-Small-Cell Lung Cancer. N Engl J Med 2005; 353:123-132.
12. Hossein Borghaei, D.O., Luis Paz-Ares, M.D. Nivolumab versus Docetaxel in Advanced Nonsquamous Non–Small-Cell Lung Cancer; CheckMate 057 N Engl J Med 2015; 373:1627-1639 October 22, 2015
13. Edward B. Garon, Naiyer A. Rizvi, Pembrolizumab for the Treatment of Non–Small-Cell Lung Cancer; KEYNOTE-001 Investigators N Engl J Med 2015; 372:2018-2028May 21, 2015.
14. http://www.esmo.org/Oncology-News/Acquired-Resistance-to-Immunotherapy?hit=mail-snews&utm_campaign=Scientific&utm_source=hs_email&utm_medium=email&utm_content=26383678.
Articole din ediţiile anterioare
Metastaze skip şi supravieţuirea - argumente pentru disecţia ganglionilor limfatici în cancerul pulmonar
Lung cancer continues to be the most common cause of death related to cancer per year and the number is increasing every year. One case out of 10 i...
De actualitate în cancerul pulmonar
Lung cancer remains the most frequent cancer in the world. For the moment we don’t have an optimal treatment especially for advanced stages. For th...