Learning objectives. To review the computed tomograpy (CT) and magnetic resonance imaging (MRI) features of hypervascular pancreatic metastases (HPM). To discuss the main differential diagnosis of HPM. Materials and method. We retrospectively analyzed images from the CT and MRI evaluations of six patients who were diagnosed with HPM by morphopathological examination in the last ten years. Results. In our study, hypervascular pancreatic metastases corresponded to renal cell carcinoma, breast carcinoma and uterine leiomyosarcoma. In non-enhanced CT (NECT), all lesions were isodense or slightly hypodense. After i.v. injection of contrast medium (CM), hypervascular pancreatic metastases have displayed an early arterial phase enhancement, followed by rapid washout in the portal and delayed phases images. Contrast-enhanced CT showed for pancreatic metastases from uterine leiomyosarcoma a gradual enhancement. The common appearance in MRI for HPM was hypointense in T1 weighted image (wi), heterogeneous aspect in T2 wi or moderately hyperintense signal with restricted diffusion. After Gd-based CM i.v. injection, HPM showed the same dynamic as in CT evaluation. Conclusions. HPM may create a diagnostic challenge, therefore the pathological examination remains essential and represents the gold standard for the final diagnosis.
Metastaze pancreatice hipervasculare: sunt un diagnostic imagistic uşor?
Hypervascular pancreatic metastasis: an easy imaging diagnostic?
First published: 28 octombrie 2020
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/OnHe.52.3.2020.3711
Abstract
Rezumat
Obiectiv. De a revizui caracteristicile computer-tomografice (CT) şi de imagistică prin rezonanţă magnetică (IRM) ale metastazelor pancreatice hipervascularizate (MPH). De a discuta diagnosticul diferenţial al MPH. Materiale şi metodă. Am analizat retrospectiv imaginile CT şi IRM a şase pacienţi care au fost diagnosticaţi prin examen morfopatologic cu metastaze pancreatice hipervascularizate în ultimii 10 ani. Rezultate. În studiul nostru, MPH au corespuns carcinomului renal, carcinomului mamar şi leiomiosarcomului uterin. În examinarea CT fără contrast, toate leziunile au fost izodense sau discret hipodense. După injectarea i.v. a contrastului, MPH au prezentat la CT o captare intensă în faza arterială precoce, urmată de spălare rapidă în imaginile din faza portală şi tardivă. Metastaza pancreatică din leiomiosarcomul uterin a prezentat o captare progresivă în evaluarea CT cu contrast. Aspectul curent al metastazelor pancreatice hipervasculare în IRM a fost hipointens în ponderaţia T1, cu semnal heterogen în ponderaţia T2 şi moderat hiperintens în ponderaţia de difuzie, dinamica postcontrast fiind similară cu cea a CT. Concluzii. Aspectul imagistic al MPH constituie o provocare pentru diagnostic, examinarea morfopatologică rămânând esenţială şi reprezentând standardul de aur pentru diagnosticul final.
Objective. To review the CT and MRI features of hypervascular pancreatic metastases (HPM) and to discuss the main differential diagnosis of HPM.
Disseminations pathways. The pancreatic metastases are divided depending on the patterns of spread: direct invasion by a primary tumor arising from adjacent organ, and hematogenous spread of metastases(1-16). The hematogenous spread of metastases to the pancreas tissue is uncommon and represents only 2% of the pancreatic malignancies. There may be a significantly long latency period between the initial diagnosis of the primary tumor and the subsequent detection of pancreatic metastases(1,2). The most common primary tumors that colonize the pancreas in studies based on surgical specimens are renal cell carcinoma, breast cancer, lung cancer, colorectal cancer and melanoma(3). Another analysis of a surgical and autopsy database showed that, in the autopsy series, the most frequent primary tumors with pancreatic metastases were represented by lung carcinoma (42%), gastrointestinal tract (24.7%) and kidney carcinomas (5%), whereas in the surgical database, there were lymphomas (29%), carcinomas of the stomach (18.4%), kidney (15.7%), malignant gastrointestinal stromal tumor (7.9%), and lung carcinoma (5.3%)(2,4).
Clinical aspects. Pancreatic metastases (PM) are asymptomatic in more than 50% of cases; these lesions are typically an incidental finding on CT performed for follow-up in oncological patients(5). The clinical symptoms produced by PM are variable and nonspecific, including abdominal pain, back pain, weight loss, nausea, melena, jaundice, gastrointestinal obstruction and upper gastrointestinal bleeding(2).
Lesion distribution and morphology. Three patterns of pancreatic metastases have been described(2): 1) multiple small nodules, which can coalesce occasionally into larger masses (5-10% of cases); 2) diffuse infiltration of the pancreas, given a global enlarged organ (15-44% of cases); 3) a solitary mass (50-75% of cases).
Materials and method. We retrospectively analyzed the imaging exams of six patients (three men and three women) who were diagnosed with HPM by morphopathological examination during a 10-year period.
CT technique. Non-enhanced CT (NECT) and contrast-enhanced CT (CECT), using currently tree phases: early arterial phase at 25-30 s after contrast material (CM) i.v.injection, pancreatic phase obtained 40-45 s after CM i.v. injection, and portal venous phase obtained 70 s after CM i.v. injection, after the injection of 1.5 ml/kg b.w. of non-ionic iodine-based CM with a power injector, flow rate 3 ml/s, and a bolus trigger/ROI into the abdominal aorta (threshold 120 UH). After the CT examination, we performed multiplanar reconstructions (MPR), maximum intensity projection reconstructions (MIP), or volumetric rendering technique (VRT).
MRI technique. We used breath hold acquisitions at a 1.5 T MR system and a body phased array abdominal coils. The protocol included three plane localizer, axial and coronal T2 ,T1 Fat Sat weighted sequences, MRCP, long and short TE SS FSE acquisition, diffusion weighted image (DWI) with multiple b values (0; 500; 800 mm/s2) correlated with ADC (apparent diffusion coefficient) map analysis, and non-enhanced and enhanced 3D FSPGR T1 Fat Sat sequence, using a dynamic multiphase acquisition, with gadolinium-based CM i.v. injection (0.1 ml/kg b.w.).
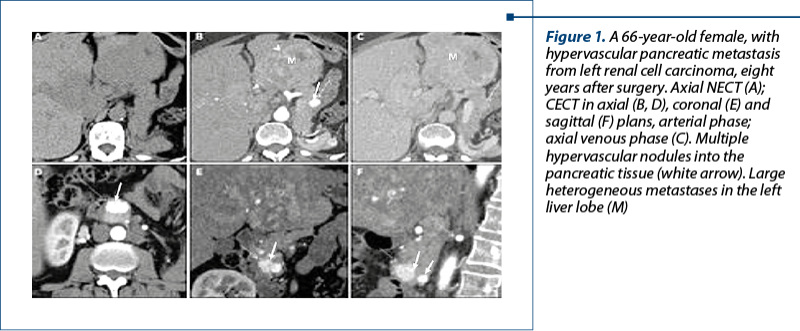
Imaging findings. In our study, hypervascular PM corresponded to: renal cell carcinoma (three cases), breast carcinoma (one case) and uterine leiomyosarcoma (one case).
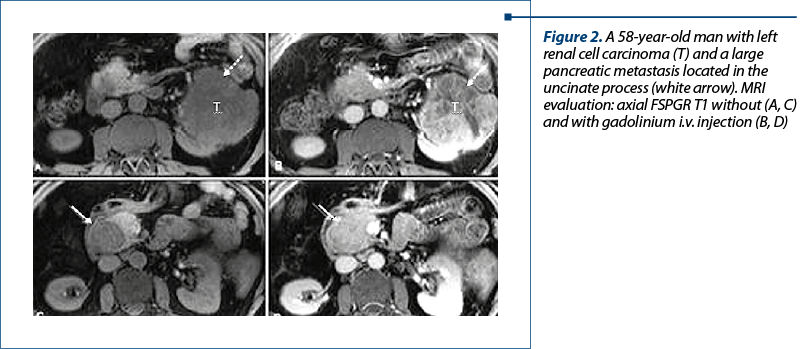
1. Renal cell carcinoma (RCC). Pancreatic metastases can be synchronous with the RCC or can appear many years after surgical procedures. The longest interval for PM appearance after RCC in our study was 22 years old. We found two patterns of PM distribution: solitary mass (one case), and multiple small nodules (two cases).
CT aspects (Figure 1). In NECT, pancreatic metastases from RCC appear as an isodense/slightly hypodense nodule (s); in CECT, pancreatic metastases showed early arterial phase enhancement, followed by rapid washout in the portal and late phase. A large mass may present heterogeneous enhancement caused by areas of necrosis.
MRI aspects (Figure 2). HPM are hypointense in T1 FS wi, with moderate hyperintense pattern in T2wi compared to the normal pancreatic parenchyma; after Gd-based CM i.v. injection HPM are hyperenhancing lesion(s), and present in DWI restricted diffusion.
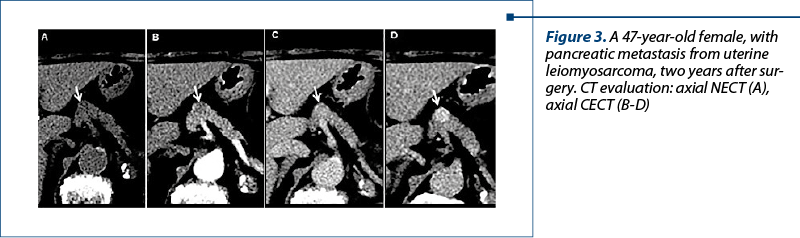
2. Uterine leiomyosarcoma (LMS). Uterine leiomyosarcoma is a rare and aggressive malignant neoplasm of the myometrial smooth muscle cells. The most common sites of distant metastases from LMS are the lung, kidney and liver(11). Metastases to the pancreas are rare(12-16).
CT aspect (Figure 3). In our case, the pancreatic metastasis corresponded to a hypodense nodule localised in the pancreatic body with a gradual enhancement in CECT.
MRI aspect. The pancreatic metastasis appears as a hypointense nodule on T1wi, with moderate hypersignal on T2wi and restricted diffusion. After Gd-based CM i.v. injection, small nodule of PM shows gradual enhancement.
3. Breast carcinoma. Breast cancer most commonly spreads to the liver, brain, adrenals, lung, ovary and bone. Metastases to the pancreas from breast cancer usually occur in the setting of diffusely metastatic multiorgan disease(14). Pancreatic metastases from breast cancer are rare, with a reported rate of 13% in an autopsy series(15).
CT aspects. In NECT, pancreatic metastases are isodense lesions with hyperattenuation in CECT compared to the normal enhanced pancreatic parenchyma.
MRI aspects. T1wi: hypointense lesion; T2wi: moderate hyperintense lesion with restricted diffusion; after Gd injection, the small metastases present a homogenous hyperenhancement, while the large lesions present a heterogeneous enhancing.
Differential diagnosis
1. Pancreatic neuroendocrine tumors (PNET). Neuroendocrine tumors of the pancreas represent 3% of all pancreatic neoplasms. The insulinoma is the most common, followed by gastrinoma, VIPoma and glucagonoma. Somatostatinoma is very rare(16-23).
Imaging findings. In general, PNETs are isodense with pancreatic parenchyma in NECT images and demonstrate avid arterial enhancement. They can be localized within the gland or have an exophytic growth. Rarely, they may cause pancreatic duct dilatation or obstruction and local vascular encasement or invasion(15).
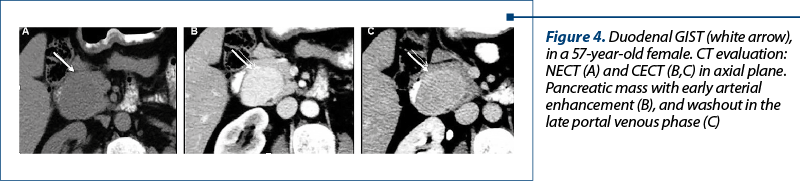
2. Invasive Duodenal GIST – CT findings. Many of these tumors arise from the muscularis propria and have an exophytic growth (Figure 4). NECT: isodense mass; homogenous early arterial enhancement for small lesions, and heterogeneous enhancing for large lesions due to necrotic areas.
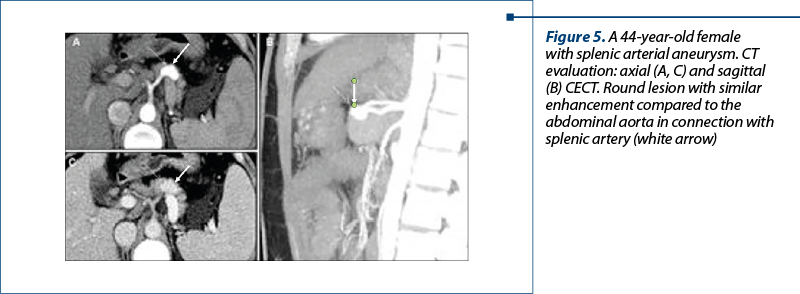
3. Vascular entities: aneurysms of the splenic artery. Peripancreatic arterial aneurysm may mimic pancreatic hypervascular lesions. The most common visceral aneurysms involve the splenic artery (60%) and the hepatic artery (20%), while mesenteric and pancreaticoduodenal arterial aneurysms are less frequent(24).
Imaging findings – CECT: an aneurysm appears as an oval or round lesion with similar enhancement with the abdominal aorta (Figure 5).
4. Intrapancreatic accessory spleen. Accessory spleen represents a congenital ectopic splenic tissue. The prevalence of intrapancreatic accessory spleen is 9.3%(24).
What should the clinician know? The description of the pancreatic lesion: localization, number, dimensions; the involvement of adjacent vessels and other anatomical structure (duodenum, extrahepatic biliary ducts). Is it a primary or a secondary pancreatic tumor? The pancreatic metastases are isolated metastases or a generalized dissemination? Is there a primary tumor recurrence associated?
Conclusions
The imaging findings in HPM may create a diagnostic challenge. The morphopathological examination is essential and represents the gold standard for the final diagnosis.
Bibliografie
-
Tan CH, et al. Imaging Features of Hematogenous Metastases to the Pancreas: Pictorial Essay. Cancer Imaging. 2011; 11: 9–15.
-
Triantopoulou C, et al. Metastatic disease to the pancreas: an imaging challenge. Insights Imaging. 2012; 3: 165-172.
-
Sweeney AD, et al. Value of pancreatic resection for cancer metstatic to the pancreas. J Surg Res. 2010; 160: 268-276.
-
Adsay NV, et al. Secondary tumours of the pancreas: an analysis of a surgical and autopsy database and review of the literature. Virchows Arch. 2010; 444: 527-535.
-
Kusnierz K, et al. Results of Surgical Management of Renal Cell Carcinoma Metastatic to the Pancreas. Contemporary Oncology. 2015; 19.1: 54–59.
-
Ahmed S, et al. Metastatic disease to the pancreas: pathologic spectrum and CT patterns. Abdom Imaging. 2013; 38: 144.
-
Merkle EM, et al. Metastases to the pancreas. Br J Radiol. 1998; 71: 1208-1214.
-
Siegelman “Body MRI”, 1st Ed. In: Elseviers Saunders. Philadelphia; 2005; 99.
-
Sperti C, et al. Metastases to the pancreas. World J Gastrointest Onco. 2014; 6 (10): 381-392.
-
Moussa A, et al. Pancreatic metastases: a multicentric study of 22 patients. Gastroenterol Clin Biol. 2004 Oct; 28(10 Pt 1):872-6.
-
Ozturk S, et al. Isolated metastasis of uterine leiomyosarcoma to the pancreas: report of a case and review of the literature. Int J Surg Case Rep. 2014; 5: 350–353.
-
Iwamoto I, et al. Metastasis of uterine leiomyosarcoma to the pancreas. J Obstet Gynaecol Res. 2005; 31: 531–534.
-
Gómez JA, et al. Uterine leiomyosarcoma metastasis to the pancreas: report of a case and review of the literature. J Gastrointest Cancer. 2012; 43: 361–363.
-
Showalter SL, Hager E, Yeo CJ. Metastatic disease to the pancreas and spleen. 2008; Department of Surgery Faculty Papers. Paper 13. Available at: https://jdc.jefferson.edu/cgi/viewcontent.cgi?article=1012&context=surgeryfp
-
Galvin A, Sutherland T, Little AF. Part 1: CT characterisation of pancreatic neoplasms: a pictorial essay. Insights into Imaging. 2011; 2(4): 379-388.
-
Ştefan F, Boroş M, Lupescu IG. What to know about imaginig aspects in pancreatic metastases. Available at: https://dx.doi.org/10.1594/ecr2017/C-1400.
-
Chawla S, Mekaroonkamol P, Willingham FF. Less common solid tumors of pancreas; Pancreatic Masses, Advances in Diagnosis and Therapy. Editors: Mihir S. Wagh, Peter V. Draganov; 978-3-319-19677-0 (Online).
-
Fesinmeyer MD, Austin MA, Li CI, De Roos AJ, Bowen DJ. Differences in survival by histologic type of pancreatic cancer. Cancer Epidemiol Biomarkers Prev. 2005; 14(7): 1766–73.
-
Vagefi PA, Razo O, Deshpande V, et al. Evolving patterns in the detection and outcomes of pancreatic neuroendocrine neoplasms: the Massachusetts General Hospital experience from 1977 to 2005. Arch Surg. 2007; 142(4): 347–54.
-
Cheema A, Weber J, Strosberg JR. Incidental detection of pancreatic neuroendocrine tumors: an analysis of incidence and outcomes. Ann Surg Oncol. 2012; 19(9): 2932–6.
-
Feldman JM. Carcinoid tumour and the carcinoid syndrome. Curr Probl Surg. 1989; 26: 835–885.
-
Maurer CA, et al. Carcinoid of the pancreas: clinical characteristics and morphological features. Eur J Cancer. 1996; 7: 1109–1116.
-
Saint-Marc O, Cogliandolo A, Pozzo A, Pidoto RR. A primary pancreatic carcinoid tumour with unusual clinical complaints: A case report. World Journal of Surgical Oncology. 2004; 2:3.
-
Vernuccio F, Borhani AA, Dioguardi Burgio M, et al. Common and uncommon pitfalls in pancreatic imaging: it is not always cancer. Abdom Radiol. 2016; 41: 283.
Articole din ediţiile anterioare
Chimioterapia citotoxică – principii şi indicaţii în cancer
Chimioterapia cancerului este tratamentul sistemic medicamentos care se bazează pe utilizarea unor substanţe care interferează cu metabolismul ...
Mai există un rol pentru nefrectomia citoreducţională în era terapiilor ţintite şi a imunoterapiei?
Renal cell carcinoma (RCC), the most frequent type of kidney cancer, responsible for 5% of oncological diagnosis in men and for 3% in women, repres...
Terapia anti HER-2 în cancerul sânului, o scurtă privire asupra opţiunilor terapeutice disponibile
În această prezentare voi detalia noile terapii disponibile pentru tratarea cancerului de sân cu supraexpresie HER-2 (o oncogenă a familiei EGFR - ...
Youth Cancer Europe Open Space Summit. Key Outcomes Report
The focus of the Open Space Summit was to assess the most urgent unmet needs that young people across Europe have identified and to enable an open ...