Gadoxetic acid, or Gd-EOB-DTPA (Primovist®), is a magnetic resonance imaging (MRI) hepatospecific T1-positive contrast material used to evaluate the hepatobiliary region and its disorders. In this article, we review the MRI protocols with Primovist®, the importance of the hepatobiliary phase, and the current clinical indications of Primovist® in oncological patients evaluated using multiparametric MRI.
Multiparametric MRI with gadoxetic acid (Primovist®) in oncological patients: current indications and utility of the hepatobiliary phase
IRM multiparametrică cu acid gadoxetic (Primovist®) la pacienţii oncologici: indicaţii curente şi utilitatea fazei hepatobiliare
First published: 26 octombrie 2023
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/OnHe.64.3.2023.8770
Abstract
Rezumat
Acidul gadoxetic, sau Gd-EOB-DTPA (Primovist®), este un agent de contrast T1-pozitiv, hepatospecific, utilizat în imagistica prin rezonanţă magnetică (IRM) pentru evaluarea regiunii şi a patologiei hepatobiliare. În acest articol, vom trece în revistă protocoalele IRM cu Primovist®, importanţa fazei hepatobiliare şi indicaţiile clinice actuale ale Primovistului® la pacienţii oncologici evaluaţi prin IRM multiparametrică.
Introduction
Gadoxetic acid, or Gd-EOB-DTPA (Primovist®), is a magnetic resonance imaging (MRI) hepatospecific T1-positive contrast material (CM) used to evaluate the hepatobiliary region and its disorders. After the vascular phase and distribution into the extravascular spaces during the dynamic MRI evaluation, gadoxetic acid is taken up progressively by hepatocytes and excreted into the bile ducts during the hepatobiliary phase (HBP). The findings obtained from the dynamic 3D T1 FatSat evaluation – in arterial phase, portal venous phase, transitional phase and HBP – are relevant to detect and characterize focal liver lesions and for the evaluation of morphology and function of the liver parenchyma and biliary tree(1-3).
MRI protocol with Primovist®
The “classical” MPMRI protocol with Primovist® includes: T2 HASTE image in coronal or axial plane, axial chemical shift image (T1 with TE in and out of phase – detection of liver steatosis and focal liver tumors with fat content), 3D T1 FatSat precontrast image, followed by i.v. slow rate injection (1 ml/s) of the CM (0.1 ml/kg body) and 20-30 ml of saline solution, using 3D T1 FatSat dynamic multiphase acquisition in late arterial phase (AP), portal venous phase (PVP) and transitional phase (TP) – appreciation of the liver mass vascularity, T2 FatSat axial image – detection of hyperintense liver lesions, diffusion and ADC map (appreciation of the intratumoral cellularity), T2 GRE (detection of hemochromatosis and siderotic nodules) and hepatobiliary phase(1-4).
Hepatobiliary phase. In patients with normal liver function, parenchymal enhancement and biliary tree opacification are optimal 20 minutes after the injection of Gd-EOB-DTPA. In patients with cholestasis, HBP must be prolonged at 40-60 minutes or even more after Primovist® i.v. injection. HBP gives information about liver function and is now accepted as a noninvasive biomarker of hepatobiliary disorders(1-3).
Abbreviated MRI protocols have the role to decrease examination times. Preliminary studies have reported good results from abbreviated Primovist®-enhanced MRI protocols in patients with suspected hepatocellular carcinoma (HCC) or colorectal cancer (CRC) metastases(4,5). In the detection of HCC, combining information from T2-weighted image (wi) and the HBP phase offer a negative predictive value similar to that obtained with conventional protocols(3,4). In the follow-up of patients with CRC, an abbreviated protocol containing T2wi, a hepatobiliary sequence, and DWI with ADC map could be a good alternative(5).
Non-enhanced abbreviated MRI protocol comprises T1- and T2-wi with DWI.
Ancillary features of HCC using non-enhanced abbreviated MRI include mild-to-moderate T2 hyperintensity, restricted diffusion, iron or fat sparing, fat in the mass, and potential blood products in the mass.
Enhanced abbreviated MRI protocol is performed after i.v. administration of the hepatobiliary CM outside de magnet, using a full dose corelated to the body weighted of the patient. After 10 minutes, the patient is positioned into the magnet and the exam starts after the three-plane localizer with T2-wi (Haste or FSE), DWI and the HBP 3D T1 FS(4). Another abbreviated enhanced MRI liver protocol is performed after i.v. administration of the hepatospecific CM outside de magnet using two-thirds of the CM dose followed by the same steps presented before, the MRI protocol ending with 3D pre-/post-CM i.v. injection in arterial phase using the remaining one-third of the hepatospecific CM dose(5).
The “mechanism” to detect HCC is the evaluation of ancillary features using the HBP: hypointensity of the nodule in association with restricted diffusion. The T2-wi with short and long TE helps to delineate between cysts, hemangiomas and HCC, compared to HBP in which all these three entities are hypointense(3).
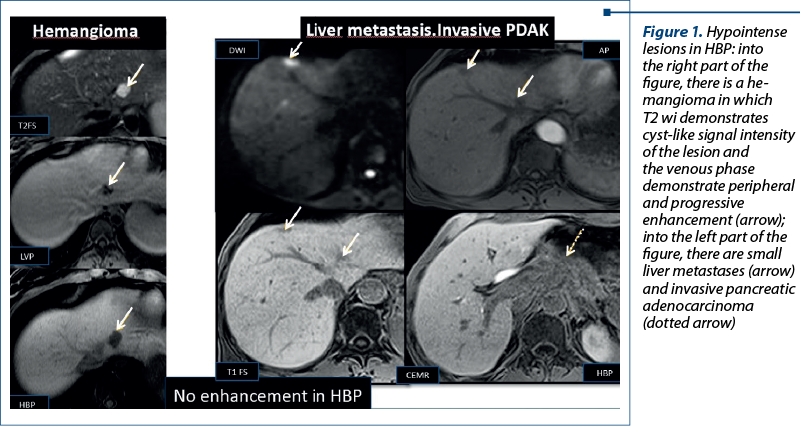
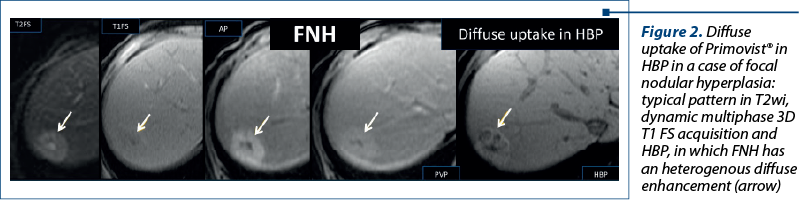
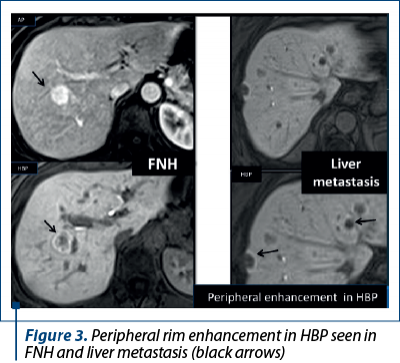
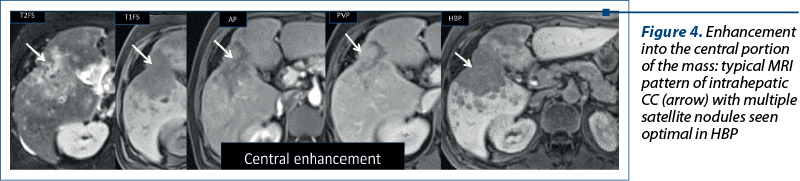
Aspects of focal liver lesions in HBP. Four patterns can be identified in HBP using Primovist®: 1) no enhancement or hypointensity; 2) diffuse uptake; 3) peripheral rim enhancement; and 4) central lesion enhancement(3).
1. No enhancement of the lesion due to the lack of expression of OATP1B3 (Figure 1). This finding can be observed in both benign (hemangiomas) and malignant lesions (liver metastases)(3).
2. Diffuse uptake of Primovist®, homogenous or heterogenous (Figure 2): the degree varies with the expression of the transporters. It is typically seen in focal nodular hyperplasia (FNH) and in FNH-like lesions, in areas of spared liver parenchyma, in patients with steatosis in some subtypes of adenomas, and in well-differentiated HCC(3,6-8).
3. Peripheral rim enhancement like in FNHs and in liver metastases (Figure 3).
4. Enhancement into the central portion of the lesions due to discrete retention of the contrast material by fibrous stroma (like in cholangiocarcinoma – Figure 4) or due to an abnormal overexpression of OATP1B3 (in some types of liver metastases)(3).
Indications of gadoxetic acid – enhanced MRI and clinical scenarios in liver pathology
The main clinical use of MPMRI with Primovist® is to detect and characterize focal liver lesions. Four clinical situations are possible: 1) to differentiate between an FNH and adenoma; 2) to detect small (<2 cm) HCCs; 3) to evaluate liver transplants; 4) to detect liver metastases(3).
FNH versus adenoma. The presence of HBP hyperintensity and polylobulated morphology were MRI specific findings in FNH diagnosis(1,3,6-9). The enhancement of FNHs is homogeneous in 23-59% of cases, and a peripheral ring of greater enhancement (owing to overexpression of OATP3B1) is seen in 23-66% of cases(8). Most adenomas do not show uptake during the HBP, showing homogeneous hypointensity signal. This aspect together with signal intensity loss using 2D dual GRE T1 acquisition with TE out-of-phase sequences, due to intracellular fat content and the slight enhancement on dynamic arterial phase, make possible the diagnostic of adenoma(3,6,10).
Hepatocellular carcinoma. In nodules measuring more than 1 cm in diameter, the combination of hyperenhancement in the late arterial phase with washout in the portal venous phase makes possible to diagnose HCC without a biopsy(2,11). This pattern occurs only in a limited number of small HCC nodules. From the molecular viewpoint, the degree of OATP1B3 expression correlates with the stages of hepatocarcinogenesis (Figure 5). When dysplastic nodule evolves into early HCC, even before neoarterialization, the expression of this polypeptide is already decreased and the lesion appears hypointense in the HBP. Based on this aspect, the guidelines from the Asian Pacific Association or the Study of the Liver, the Japan Society of Hepatology, and the Korean Liver Cancer Association – National Cancer Center have extended the definition of washout to the HBP(3). Atypical HCC has the following characteristics: wash in AP, no wash out in PVP, hypointense in HBP. Early HCC has no wash in into the AP, but presents washout in PVP, is hypointense in HBP, and has restriction in DWI(2,11-14).
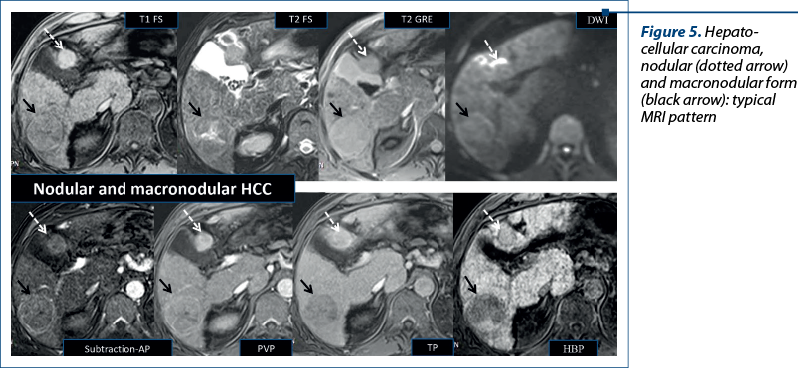
It is important to remember that 10% of HCCs are hyperintense in the HBP due to the overexpression of OATP1B3(8,9), this aspect indicating a lower aggressivity and a better prognosis(3).
Together with other MRI findings, the enhancement aspect of the liver nodule into the HBP can be a useful biomarker for measuring the risk of developing HCC(3).
Primovist®-enhanced MRI provides accurate staging of HCC before and after treatment, with locoregional/biologic therapies, including even microvascular invasion, and is also considered to predict the recurrence of HCC after liver transplantation(13,14,16,17).
Detection of liver metastases. The combination of HBP images and diffusion-wi is better than other techniques for detecting small metastasis(12,14). Primovist®-enhanced MRI plays an important role in the diagnosis of liver involvement in patients with CRC(5). Its sensitivity for detecting liver metastases measuring less than 1 cm is 93%, compared with 42% for CT(15). The enhancement pattern of metastases in the HBP is correlated with the prognosis or survival. Liver lesions that were homogeneously hypointense in the HBP were associated with a longer survival than the mixed lesions(3).
Conclusions
In current clinical practice, for oncological patients, Primovist®-enhanced MPMRI offers information about the structure and function of the liver parenchyma and biliary tree, in a single examination, improving the detection and characterization of focal liver lesions, which are very important for the management of oncological patients, allowing a noninvasive assessment of liver function.
Conflict of interest: none declared.
Financial support: none declared.
This work is permanently accessible online free of charge and published under the CC-BY licence.

Bibliografie
-
Ringe KI, Husarik DB, Sirlin CB, Merkle EM. Gadoxetate disodium-enhanced MRI of the liver: Part 1. Protocol optimization and lesion appearance in the noncirrhotic liver. AJR Am J Roentgenol. 2010;195(1):13–28.
-
Cruite I, Schroeder M, Merkle EM, Sirlin CB. Gadoxetate disodium-enhanced MRI of the liver: Part 2. Protocol optimization and lesion appearance in the cirrhotic liver. AJR Am J Roentgenol. 2010;195(1):29–41.
-
Baleato-Gonzalez S, Vilanova JC, Luna A, et al. Current and Advanced Applications of Gadoxetic Acid-enhanced MRI in Hepatobiliary Disorders. Radiographics. 2023;43(4):2-21.
-
Park HJ, Kim SY, Singal AG, et al. Abbreviated magnetic resonance imaging vs ultrasound for surveillance of hepatocellular carcinoma in high-risk patients. Liver Int. 2022;42(9):2080–2092.
-
Ghorra C, Pommier R, Piveteau A, et al. The diagnostic performance of a simulated “short” gadoxetic acid-enhanced MRI protocol is similar to that of a conventional protocol for the detection of colorectal liver metastases. Eur Radiol. 2021;31(4):2451–2460.
-
McInnes MD, Hibbert RM, Inacio JR, Schieda N. Focal Nodular Hyperplasia and Hepatocellular Adenoma: Accuracy of Gadoxetic Acid-enhanced MR Imaging – A Systematic Review. Radiology. 2015;277(2):413–423.
-
Bilreiro C, Soler JC, Ayuso JR, Caseiro-Alves F, Ayuso C. Diagnostic value of morphological enhancement patterns in the hepatobiliary phase of gadoxetic acid-enhanced MRI to distinguish focal nodular hyperplasia from hepatocellular adenoma. Radiol Med (Torino). 2021;126(11):1379–1387.
-
Vernuccio F, Gagliano DS, Cannella R, Ba-Ssalamah A, Tang A, Brancatelli G. Spectrum of liver lesions hyperintense on hepatobiliary phase: an approach by clinical setting. Insights Imaging. 2021;12(1):8.
-
Fujita N, Nishie A, Asayama Y, et al. Hyperintense Liver Masses at Hepatobiliary Phase Gadoxetic Acid-enhanced MRI: Imaging Appearances and Clinical Importance. RadioGraphics. 2020;40(1):72–94
-
Yoneda N, Matsui O, Kitao A, et al. Benign Hepatocellular Nodules: Hepatobiliary Phase of Gadoxetic Acid-enhanced MR Imaging Based on Molecular Background. RadioGraphics. 2016;36(7):2010–2027.
-
Choi JW, Lee JM, Kim SJ, et al. Hepatocellular carcinoma: imaging patterns on gadoxetic acid-enhanced MR Images and their value as an imaging biomarker. Radiology. 2013;267(3):776–786.
-
Park SH, Kim H, Kim EK, et al. Aberrant expression of OATP1B3 in colorectal cancer liver metastases and its clinical implication on gadoxetic acid-enhanced MRI. Oncotarget. 2017;8(41):71012–71023.
-
Ayuso C, Forner A, Darnell A, et al. Prospective evaluation of gadoxetic acid magnetic resonance for the diagnosis of hepatocellular carcinoma in newly detected nodules ≤2 cm in cirrhosis. Liver Int. 2019;39(7):1281–1291.
-
Koh DM, Ba-Ssalamah A, Brancatelli G, et al. Consensus report from the 9th International Forum for Liver Magnetic Resonance Imaging: applications of gadoxetic acid-enhanced imaging. Eur Radiol. 2021;31(8):5615-5628.
-
Vilgrain V, Esvan M, Ronot M, Caumont-Prim A, Aube C, Chatellier G. A meta-analysis of diffusion-weighted and gadoxetic acid-enhanced MR imaging for the detection of liver metastases. Eur Radiol. 2016;26(12):4595–4615.
-
Yang H, Han P, Huang M, et al. The role of gadoxetic acid-enhanced MRI features for predicting microvascular invasion in patients with hepatocellular carcinoma. Abdom Radiol (NY). 2022;47(3):948–956.
-
Lee S, Kim KW, Jeong WK, et al. Gadoxetic acid-enhanced MRI as a predictor of recurrence of HCC after liver transplantation. Eur Radiol. 2020;30(2):987–995.
Articole din ediţiile anterioare
Imagistica multiparametrică de rezonanţă magnetică în diagnosticul cancerului de prostată: o necesitate
Cancerul de prostată reprezintă o importantă problemă de sănătate publică. Imagistica prin rezonanţă magnetică (IRM) este cea mai bună modalitate d...
Evaluarea IRM multiparametrică în hiperplazia benignă de prostată – o adevărată provocare în excluderea cancerului de prostată
Evaluarea IRM multiparametrică este un instrument de nepreţuit în depistarea, localizarea şi caracterizarea leziunilor de prostată, una dintre ...