Cancer is known as a considerable disease with high morbidity and mortality within adult’s populations. There are different types of cancers with a wide range of distributions and risk factors worldwide. Vaginal cancers are limited but may lead to high morbidity and mortality. Vaginal cancers have several predisposing factors, but HPV is recognized as the most important viral causative agent for vaginal cancers. There are different types of diagnostics for detecting HPV vaginal cancers. A qualified Pap smear is recognized as an invaluable screening program for early detection of HPV vaginal cancers. The incidence of vaginal cancers has significantly decreased in industrialized countries because of qualified performance of Pap smear. The most morbidity and mortality relating to vaginal cancers are reported from developing countries. There are different types of treating procedures including cryotherapy, laser therapy, electrosurgical procedures, radiotherapy and pharmacotherapy. The result of treatments in patients is checked via molecular diagnostic techniques which are known as gold standard approaches. Limitation of risk factors and application of vaccines are confirmed as favor preventing methods. The aim of this article is to review the subtitle of vaginal cancer staging, epidemiology of HPV vaginal cancers, viral characteristics and pathogenesis of HPV vaginal cancers, clinical demonstrations of HPV vaginal cancers diagnosis, treatment and prevention.
Tipuri de cancer vaginal şi HPV
Vaginal cancers and human papilloma virus
First published: 24 martie 2015
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/OnHe.30.1.2015.4301
Abstract
Rezumat
Cancerul este cunoscut ca o boală gravă cu grad înalt al morbidităţii şi mortalităţii în rândul populaţiei adulte. Există diferite tipuri de cancer cu o gamă largă a distribuţiei şi factorilor de risc la nivel mondial. Tipurile de cancer vaginale sunt limitate, dar pot duce la morbiditate şi mortalitate. Cancerul vaginal are mai mulţi factori predispozanţi, dar HPV este recunoscut ca cel mai important agent cauzal viral. Există diferite tipuri de diagnosticare pentru detectarea HPV în cancerul vaginal. Un test Papanicolau calificat este recunoscut ca un program de screening nepreţuit pentru depistarea precoce a HPV în cancerul vaginal. Incidenţa cancerelor vaginale a scăzut semnificativ în ţările industrializate datorită performanţelor testului Papanicolau. Rata morbidităţii şi mortalităţii legată de tipurile de cancer vaginal sunt raportate din ţările în curs de dezvoltare. Există diferite tipuri de proceduri de tratament inclusiv crioterapie, terapie cu laser, proceduri electrochirurgicale, radioterapie şi farmacoterapie. Rezultatul tratamentelor la pacienţi este verificat prin tehnicile de diagnosticare moleculară care sunt cunoscute ca abordări standard de aur. Limitarea factorilor de risc şi aplicarea de vaccinuri sunt confirmate ca metode favorite de prevenire. Scopul acestui articol este de a revizui subtitlul stadializarea cancerului vaginal, epidemiologia de HPV în cancerul vaginal, caracteristicile virale şi patogeneza, demonstraţii clinice în diagnosticarea HPV în tipurile de cancer vaginal, tratament şi prevenire.
Introduction
Cancer is known as one of the most important death causes among adults’ populations. About 6 million patients with cancer die every year worldwide. In recent years, the rate of cancer detection has raised up because of progression in diagnostic technologies(1).
Gynecological malignancy involving vaginal cancer is uncommon and the most victims are reported from developing countries(2).
In toto, the global portion of vaginal cancer as a gynecologic malignancy is about 3%(3).
There are different risk factors including smoking, poor hygiene, high parity, intimating with several sexual partners, in utero exposed diethylstilbestrol, immunodeficiency, genital warts and sexually transmitted Human Papilloma Virus (HPV) that predispose vaginal cancer. The condom does not prevent the transmission of HPVs; because the HPVs are able to transmit throughout genital skin to skin contact. According to recorded reports, 40% of vaginal cancers occur by HPV infections(1,4-8).
The HPV not only in women but also in men may cause genital cancers. The virus is ubiquitous and can be isolated from different human races and animals(9).
The earlier recognition of vaginal cancer was introduced in 1952 by Meigs and Graham. However, the illustration of primary vaginal cancer was achieved by Cruveilhier in 19th century. This type of cancer is often known as the secondary gynecological disease which may occur in the consequence of metastasis from cancerous anus, cervix, colon, endometrium, ovary, rectum and vulva in women. Thus, the primary form of vaginal cancer is rare(6,10,11).
There are different types of primary vaginal cancers (Table 1)(10).
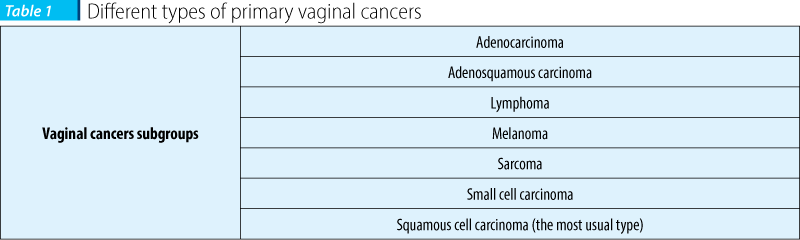
The previous reports show that squamous cell carcinoma ranks as the most prevalent vaginal cancer with the percentage of about 80. The majority of squamous cell carcinoma is in association with HPV(6).
Several investigations indicate that vaginal cancers are common among aged women with more than 60 years old. 50% of the patients with vaginal cancers are over 70 years old. The average age of mortality for patients with vaginal cancer is recorded about 65 years old(6,10).
Younger women with the age of 40 or less, involve about 10% of the population with vaginal cancer which in most cases caused by HPV. There is a direct association between HPV and cervico-vaginal cancer. In utero contact diethylstilbestrol is the predominant cause of vaginal cancers of melanoma and adenocarcinoma(3,6,10).
HPV family includes more than 100 carcinogenic and non-carcinogenic types. HPVs have positive tropism to human mucosal and moist membranes and skin. However, between 30%-40% of HPV genotypes may lead to human genital tract. The low risk carcinogenic types of HPVs are important causative agent for viral warts such as genital warts (Table 2)(3,9,12).

The aim of this article is to review the subtitle of vaginal cancer staging, epidemiology of HPV vaginal cancers, viral characteristics and pathogenesis of HPV vaginal cancers, clinical demonstrations of HPV vaginal cancers diagnosis, treatment and prevention.
Vaginal cancer staging
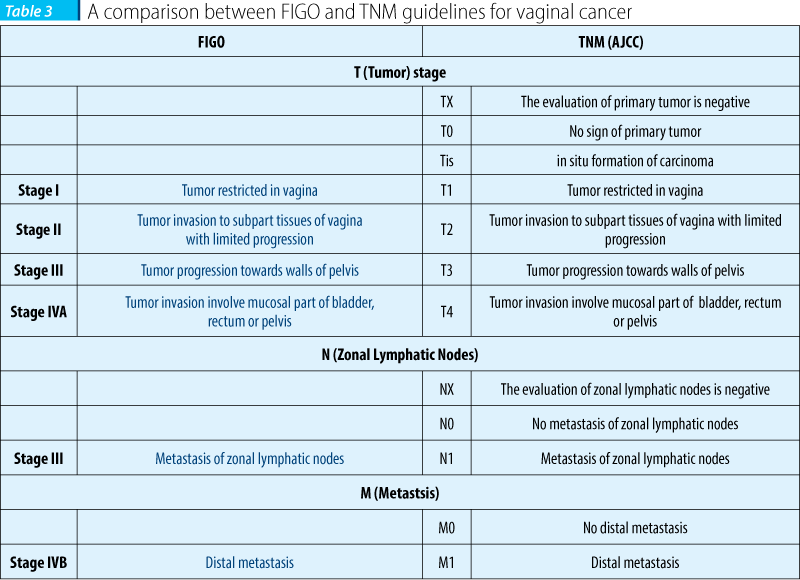
There are two types of guidelines regarding to vaginal cancer staging systems involving the International Federation of Gynecology and Obstetrics (FIGO [http://www.figo.org/]) and the American Joint Commission on Cancer ((AJCC [https://cancerstaging.org/]) (TNM (Tumor-Lymph Nodes-Metastasis) staging system)(13,14).
FIGO and AJCC have different views on vaginal cancer staging. The FIGO staging has a clinical attitude, while the AJCC’s attitude is on the basis of surgical methodology(13).
Table 3 has a comparable attitude on FIGO and AJCC guidelines classifications for vaginal cancer(13,14).
Epidemiology of HPV vaginal cancers
HPV causes a wide range of cervical and non-cervical cancers. Non-cervical cancers involve anal, oropharyngeal, penile, vaginal and vulvar ones. The incidence of HPV vaginal cancers is considerably high in developing countries and leads to a high rate of mortality among women. According to previous researches, the screening programs relating to vaginal cancers have led to a significant reduction of incidence and deaths in industrialized countries in comparison with developing countries(15).
The HPV is the most common viral agent that causes sexually transmitted diseases. The asymptomatic HPV infections vary from 2% to 44%. Despite ranging a diverse number of HPV carcinogenic types, HPV 16 (~60%) and HPV 18 (10%) are known as the most important cervico-vaginal cancer around the world. It is estimated that 80% of 50 years old women and elder are infected by HPV causing STD(8,16).
A low number of carcinogenic types of HPVs may trigger the formation of malignant carcinomas and tumors in vagina. Furthermore, the severity of multi-genotype HPV (2-5 types) infections is much more than monotype HPV infections(9,17).
Viral characteristics and pathogenesis
of HPV vaginal cancers
Vagina encompasses an expanded tubular structure with a length of 7-8 cm in size and a triple layered wall. The vagina wall is made up of a three-layered structure which involves mucosa, muscularis and adventitia respectively. The inner side of vagina is covered by stratified and non-keratinized squamous epithelial tissues(14).
HPVs cause different types of epithelial injuries with a wide range of characteristics. They are able to attack to either cutaneous or mucosal tissues. The HPVs which attack to mucosal tissues may induce vaginal cancers in human being hosts(9,18).
HPV is a 55 nm particle in diameter which possesses a 72 capsomeric icosahedral capsid and classified as a circular double stranded DNA viral agent with no envelope. HPV is a member of Papillomaviridae family. The active virion encompasses a genome encoding both of early (E1, E2, E4-E7) and structural (L1 and L2) genes. The presence of the genes empowers HPV to replicate, transcript and oncogenesis. In the most cases (70%-90%), the genital infections caused by HPV disappear in a period of 1-2 years; but the persistence of HPV may lead to cervical and non-cervical (e.g. vaginal) cancers(9,16).
Several studies show a series of tissue injuries in consequence of HPV infection which may not to appear cancers. Because of different viral protein productions, the type of lesions may vary. The viral proteins will be absorbed via superficial epithelial layers(18).
The persistence of high risk HPV types including 16 and 18 in genital system may lead to epithelial squamous basal cells proliferation. The other cells are resistant to HPVs. The defect of immune system and the presence of risk factors all together predispose the induction of cancer. Nearly 100%, the HPV DNA molecules are observed in clinical samples(9,16).
The high risk HPV proteins including E6 and E7 are oncogenic particles which inactivate the tumor suppression mechanism of retinoblastoma protein (pRB) and p53 within the infected cervico-vaginal epithelial cells in which the cellular transformation occurs. pRB and p53 are two important proteins that regulate the growth of host cells(9,17).
On the other hand, the E5 viral protein enhances the rate of growth and differentiation in infected vaginal epithelial cells. Viral early proteins cause a continued cell proliferation which may lead to appearance of vaginal carcinomas or tumors. The carcinogenic HPV types change the genetic characterizations of epithelial cells to form carcinoma which is noticeable for pathological distinguishes and recognitions. The cytological observations of HPV vaginal lesions indicate the presence of extrachromosomal viral DNA molecules within the host cells’ nuclei(9,17).
By the time, the more content of damaged DNA will be accumulated within the infected host cells. The accumulation of damaged DNA may lead to cell transformation and progression of cancerous growth among vaginal epithelial cells. Simultaneously, the high capacity of viral E7 proteins within the infected cells induces immortalization of the vaginal host cells. The process of vaginal cancers takes time from 2 to 20 years in women(9,17).
Clinical demonstrations
of HPV vaginal cancers
There are three forms of vaginal infections caused by HPVs(9):
a) The asymptomatic form of infection will be resolved spontaneously by the time. This kind of infection has no danger or discomfortibility for patient.
b) The inactive or latent infection with pale symptoms. The most patients are not aware from their infection. The cytology of the infected cells is normal.
c) The active and severe infection which may lead to noticeable changes within infected cells of vagina causing vaginal intraepithelial neoplasia (VAIN). The continuous of vaginal intraepithelial neoplasia will result in vaginal cancer. The HPV DNA molecules are detectable in deformed infected cells.
Diagnosis
The HPVs are not culturable via routine laboratory approaches. Also, the immunologic tests are not reliable. Thus, cytological, histological and molecular diagnostics are appropriate approaches for detecting HPV in clinical samples(3,9,18).
Cytological diagnosis
The most routine diagnostic method for detecting carcinogenic types of HPVs is Papanicolaou-stained (Pap) smear. Pap smear was introduced by George Papanicolaou in 1949 for screening cervico-vaginal cancers. The screening method of Pap smear has considerably reduced the rate of cervico-vaginal cancers and the related mortality. The qualified Pap smear screening test leads to early detection of 90% of invasive cervico-vaginal cancers. Detection of cervico-vaginal cancers at early stages prevents the progression of malignancy and helps to treat the pre-invasive lesions. The guideline updated by the American Cancer Society (ACS) in 2002 supported the early diagnosis of cervico-vaginal cancers(8,9,19).
The Pap smear method has modified over the time and now is known as Bethesda System. According to Bethesda system, there are four types of cell abnormalities including atypical squamous cells (ASC), Low-level squamous intraepithelial lesions (LSIL), High-level squamous intraepithelial lesions (HSIL) and Squamous cell carcinoma (SCC). There are some limitations with Pap smear including false negative and false positive results. The accuracy of Pap smear results is estimated about 80%. Each Pap smear slide is consisted of 50,000-300,000 cervico-vagina cells which may confuse and overwhelm the clinicians and this problem may reduce the accuracy of the technique. Several investigations corroborate that the quality of Pap smear screening is very important. In countries that perform high quality Pap smear screening tests, the incidence of cervico-vaginal cancers has decreased significantly. Naturally, the rate of morbidity and mortality pertaining to cervico-vaginal cancer has reduced too(8,9,20).
For reducing the rate of Pap smear false negative and false positive outcomes, the modified methods of PrepStain system and ThinPrep Pap smear are developed. In these techniques the uniform monolayer is prepared via cervico-vagina brush containing abundance of epithelial cells. The provided cells are fixed via preservative solutions prior to direct microscopy observation on a glass slide. The solution digests bacteria, mucus, proteins, red blood cells and yeasts; so, it cleans up the samples. There are some steps to complete provided specimens in PrepStain system and ThinPrep Pap smear. Although the costs of the two recent diagnostic methods have increased but the accuracy of the results has significantly improved(9).
Histopathological diagnosis
The Pap smear positive samples with no significant cervico-vaginal lesions must be checked via colposcopy and biopsy. The colposcopy helps to find any type of macrodysplasia within the cervico-vaginal tissues; but not microdysplasia. If the colposcopy was not able to find any abnormalities, biopsy may be a good choice to study any pathologic effects related to HPV infections including acanthosis and koilocytosis(8,9).
Molecular diagnosis
In situ hybridization (ISH) is an appropriate nucleic acid based technology for detecting HPV DNA or RNA within cervico-vaginal tissue specimens suspected for cancer or any other malignancies(9,21).
According to previous investigations, molecular diagnostic approaches are known as the gold standard methods for detecting HPV DNA in biopsies and tissue samples. In addition to ISH assay, Southern transfer hybridization (STH) and dot blot hybridization (DBH) are recognized as non-amplified hybridization molecular tests with qualified sensitivity and specificity. Hybrid capture test and Polymerase chain reaction (PCR) are other suitable molecular techniques for detecting HPV DNA molecules. Among a wide range of molecular diagnostics, PCR is recognized as the most reliable technique with high sensitivity and specificity(22).
Treatment and Prevention
As mentioned before, healthy immune systems will dominate HPVs up to 2 years and the transient transformed cells will be replaced by normal cells. In parallel with HPV cervico-vaginal infections, the local level of IgA and IgG increases but the clearance of HPVs is not achieved. However, the IgA which systemically and specifically is produced against HPVs has direct effect on virus clearance. Therefore, the majority of patients with healthy immune system are cured spontaneously. Any risk factor together with defective immune system are counted as predisposing items for progression and development of the malignancies such as cervico-vaginal cancers(9,23).
There are some alternatives including cryotherapy, laser therapy and electrosurgical procedures which can be used as suitable choices for HPV vaginal infections treatment. The cryotherapy method is used for freezing cervico-vagina tissue lesions and the peripheral area involving 5 mm to eradicate the occurred malignancy or cancer. Obviously, freezing and thawing the related tissues must be repeated for several times to induce the apoptosis mechanism within the host cells(9).
Laser therapy is as proper as cryotherapy even with more quality of healing and comfort; but more expensive than the cryotherapy. The electrosurgical procedure is as effective as laser therapy with cheaper cost(9).
The cervico-vaginal carcinomas which include tumors and proliferated tissues may be treated via radiotherapy. Radiotherapy is very influent and powerful procedure because of the high level energy of radiobeams. The radiotherapy is an appropriate method for destroying malignancies occurred in peripheral zones of lymph nodes in primary stages of tumor formations or even for advanced forms of cervico-vaginal cancers(9,10).
Among patients with different types of HPV infections and therapies, the recorded reports indicate several successful treatments and many recurrent cervico-vaginal infections, malignancies and carcinomas. The recurrent diseases are observed mostly among patients with defective immune systems. Thus, molecular techniques are known as gold standard methods for checking the accuracy of treatment. The absence of HPV DNA molecules within tissue specimens confirms a definite treatment, while the presence of the DNA rejects the accuracy of the treating method(9,18).
In addition to aforementioned therapies, chemotherapy is a general method for treating all the patients with different stages of malignancy. For example, cidofovir, a drug with wide range efficacy against HPV, may be used for proliferating inhibition within cancerous cervico-vaginal tissues. The topical use of podophyllin/vidarabine in patients may lead to eradicate the mild and moderate cervico-vaginal infections for the most(9).
For preventing HPV infections may either reduce the efficacy of some environmental risk factors or use the recombinant vaccines. The HPV vaccines have type specific activity against high risk types including HPV 16 and 18. The use of HPV vaccines is an appropriate choice for preventing cervico-vaginal malignancies caused by HPVs(3,9,22).
Conclusion
The vaginal cancers are diverse in forms, risk factors and stages. The HPV is one of the most important microbial agents causing STD such as vaginal lesions/malignancy/cancers. According to recorded reports, there are several predisposing factors which may facilitate the appearance of HPV vaginal cancers in different range of age among women populations.
The reduction of HPV vaginal cancer is easy and simple throughout the application of high qualified diagnostic techniques such as Pap smear and molecular assays.
There are several qualified treating procedures which must be determined by physicians - depending on the situation of the infection. It is necessary to check patients who are undergone treatment procedures via molecular approaches. Molecular diagnostic techniques confirm definite treatment or recurrent cases by the absence or presence of viral DNA molecules within tissue samples.
In addition to high quality diagnostic methods, decreasing risk factors (if it is possible) and using vaccines are appropriate and cost-effective criteria for preventing HPV vaginal cancers.
Bibliografie
2. Okolo CA, Odubanjo MO, Awolude OA, Akang EE. A Review of Vulvar and Vaginal Cancers in Ibadan, Nigeria. North American Journal of Medicine and Science April. 2013;6(2):77.
3. Diaz ML. Prevention of cervical, vaginal, and vulval cancers: role of the quadrivalent human papillomavirus (6, 11, 16, 18) recombinant vaccine. International journal of women’s health. 2009;1:119.
4. Wu X, Matanoski G, Chen VW, Saraiya M, Coughlin SS, King JB, et al. Descriptive epidemiology of vaginal cancer incidence and survival by race, ethnicity, and age in the United States. Cancer. 2008;113(S10):2873-82.
5. Dhakal HP, Pradhan M. Histological pattern of gynecological cancers. Journal of Nepal Medical Association. 2009;48(176).
6. Shah CA, Goff BA, Lowe K, Peters III WA, Li CI. Factors affecting risk of mortality in women with vaginal cancer. Obstetrics and gynecology. 2009;113(5):1038.
7. Blomberg M, Friis S, Munk C, Bautz A, Kjaer SK. Genital warts and risk of cancer: a Danish study of nearly 50 000 patients with genital warts. Journal of Infectious Diseases. 2012;205(10):1544-53.
8. Saslow D, Solomon D, Lawson HW, Killackey M, Kulasingam SL, Cain J, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA: a cancer journal for clinicians. 2012;62(3):147-72.
9. Burd EM. Human papillomavirus and cervical cancer. Clinical microbiology reviews. 2003;16(1):1-17.
10. Murakami N, Kasamatsu T, Sumi M, Yoshimura R, Takahashi K, Inaba K, et al. Radiation therapy for primary vaginal carcinoma. Journal of radiation research. 2013:rrt028.
11. Tewari KS, Cappuccini F, Puthawala AA, Kuo JV, Burger RA, Monk BJ, et al. Primary invasive carcinoma of the vagina. Cancer. 2001;91(4):758-70.
12. Sushma M, Vamsikrishna B, Babu M, Mohanraj R. A Review on Role of Human Papilomma Virus (HPV) In Cervical Cancer. PharmaTutor. 2014;2(6):21-30.
13. Jewell EL, Cormier B. Vaginal Cancer Staging2013. Available from: http://emedicine.medscape.com/article/2156399-overview.
14. Cardenes HR, Zook JE, Andolino DL. Carcinoma of the Vagina. Technical Basis of Radiation Therapy: Springer; 2012. p. 917-47.
15. Lowy DR, Schiller JT. Reducing HPV-associated cancer globally. Cancer Prevention Research. 2012;5(1):18-23.
16. Steben M, Duarte-Franco E. Human papillomavirus infection: epidemiology and pathophysiology. Gynecologic oncology. 2007;107(2):S2-S5.
17. Münger K, Baldwin A, Edwards KM, Hayakawa H, Nguyen CL, Owens M, et al. Mechanisms of human papillomavirus-induced oncogenesis. Journal of virology. 2004;78(21):11451-60.
18. Doorbar J. Molecular biology of human papillomavirus infection and cervical cancer. Clinical science. 2006;110:525-41.
19. Roberts JN, Kines RC, Katki HA, Lowy DR, Schiller JT. Effect of Pap smear collection and carrageenan on cervicovaginal human papillomavirus-16 infection in a rhesus macaque model. Journal of the National Cancer Institute. 2011.
20. Verma I, Jain V, Kaur T. Application of Bethesda System for Cervical Cytology in Unhealthy Cervix. Journal of clinical and diagnostic research: JCDR. 2014;8(9):OC26.
21. Zhang W, Kapadia M, Sugarman M, Free H, Upchurch C, Gniewek R, et al. Adjunctive HPV In-Situ Hybridization (ISH) Assay as an Aid in the Diagnosis of Cervical Intraepithelial Neoplasia in Cervical Tissue Specimens: An Analytical and Functional Characterization. International Journal of Gynecologic Pathology. 2012;31(6):588-95.
22. Mammas IN, Sourvinos G, Zaravinos A, Spandidos DA. Vaccination against human papilloma virus (HPV): epidemiological evidence of HPV in non-genital cancers. Pathology & Oncology Research. 2011;17(1):103-19.
23. Chua KL, Hjerpe A. Persistence of human papillomavirus (HPV) infections preceding cervical carcinoma. Cancer. 1996;77(1):121-7.