Splenic abscess in children is a rare entity and it usually occurs after local dissemination or hematogenous spread, especially in immunocompromised patients. Because of its rarity and nonspecific symptoms and signs, the splenic abscess is difficult to diagnose. We report the case of a 14-month-old girl with splenic abscesses. She was admitted to the hospital for persistent high fever, aphthous lesions and an ulcerative malleolar lesion. The laboratory tests showed leukocytosis, anemia, thrombocytosis, elevated C-reactive protein and D-dimers levels, with normal procalcitonin levels. The blood culture and the culture from the lesion were negative. The abdominal ultrasound showed two hypoechogenic lesions in the spleen. She was successfully treated with wide-broad antibiotic therapy for six weeks, with a total resolution of the lesions. The prolonged antibiotic therapy and hospitalization can lead to complications such as dysbiosis and superficial vein thrombosis. This case illustrates that splenic abscess should be considered in unknown-focus fever, even in infants with no underlying conditions. Conservative therapy should be the first-line therapy for splenic abscesses in children, considering the importance of the immunological functions of the organ.
Abcesul splenic – cauză neobişnuită de febră la sugar
Splenic abscess – unusual cause of fever in an infant
First published: 31 octombrie 2022
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Pedi.67.3.2022.7237
Abstract
Rezumat
Abcesul splenic este o entitate rară în patologia pediatrică. Acesta apare în urma diseminării locale sau hematogene a agentului microbian, în special la pacienţii imunocompromişi, splina fiind un organ limfoid. Fiind o patologie rară, cu semne şi simptome nespecifice, diagnosticul abcesului splenic se realizează cu dificultate. Prezentăm cazul unei fetiţe în vârstă de 14 luni, diagnosticată cu abces splenic. Aceasta a fost internată pentru febră înaltă persistentă, afte bucale şi o leziune ulcerativă maleolară. Investigaţiile de laborator au evidenţiat leucocitoză, anemie, trombocitoză, proteină C reactivă mult crescută, nivel crescut al D-dimerilor, dar cu un nivel normal al procalcitoninei. Hemocultura şi cultura din plagă au fost negative. Ecografia abdominală a pus în evidenţă două leziuni hipoecogene la nivelul splinei. Nu s-a pus în evidenţă un sindrom de imunodeficienţă în acest caz. Pacienta a fost tratată cu antibioterapie cu spectru larg timp de şase săptămâni, cu rezoluţia completă a leziunilor splenice. Tratamentul antibiotic prelungit şi spitalizarea de lungă durată au condus la complicaţii precum disbioză şi tromboză venoasă superficială. În cazul febrei fără focar, abcesul splenic poate fi un diagnostic posibil, chiar şi în cazul pacienţilor fără comorbidităţi. Terapia conservatoare ar trebui să fie prima linie de tratament în cazul abceselor splenice la copii, având în vedere funcţia imunologică a acestui organ.
Introduction
Splenic abscesses are a rare and uncommon medical condition, especially in the pediatric population, with a high mortality rate of around 50%(1). The exact incidence of the disease is not known, but it is considered to be increasing. Until 2004, around 600 cases of splenic abscesses in adults and children were reported(2,3).
The predisposing factors for this entity are metastatic infections from other body sites, direct extension from local infections or secondary infections arising from splenic trauma, or other conditions that might affect the structure of the spleen (hemoglobinopathies, vascular malformities, vasculitis or trauma). They are found more frequently in immunosuppressed patients(1,4,5).
The diagnosis of the abscesses is challenging, because of the unspecific symptoms. One-third of the patients present the triad of fever, left upper quadrant tenderness or abdominal pain and leukocytosis(4,5). The laboratory tests also reveal elevated erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP). The diagnosis is established based on imaging techniques. Ultrasonography (US) demonstrates the presence of the abscesses as hypoechogenic areas. The gold standard for diagnosis is represented by computed tomography (CT)(5).
Splenic abscesses are treated with broad-spectrum antibiotics. Once a microbiologic diagnosis is made, the antibiotic spectrum is narrowed. The ideal treatment is represented by drainage, resection, or splenectomy if required. Ultrasonic or CT-guided percutaneous aspiration has become the preferred alternative to splenectomy in the pediatric age group. If multiple small lesions are present, a course of prolonged antibiotic therapy may be preferred(4).
Early recognition and treatment are required, although the diagnosis can be easily missed because of the nonspecific signs and symptoms. The availability of imaging techniques leads to the early detection of abscesses and improves patient’s outcomes. The conservative treatment is preferred, especially for children, avoiding the overwhelming post-splenectomy infections syndrome (OPSI)(6,7).
Case report
We report the case of a 14-month-old girl transferred to our hospital for further investigations and for the treatment of a possible immune deficiency.
The familial medical history was unremarkable, with no immune deficiency. From the patient’s personal medical history, we notice that she is the second child of the parents, born at 40 weeks, with normal weight and length for gestational age and with an Apgar score of 10. She was a healthy child until admission, with few infections. In the past, she was in contact with her parents while being infected with SARS-CoV-2.
The disease had started four weeks before the admission to our hospital, with fever (the maximum value was 38.3ºC), which persisted despite the antipyretics’ administration. In evolution, she got sleepy, with a lack of interest in feeding and presented oral aphthae and a vesicular lesion on the medial malleolus, which became ulcerative (Figure 1). The treatment with amoxicillin/clavulanic acid and local bacitracin was initiated. Her evolution was stationary, so the girl’s admission to the Clinical Hospital for Infectious Diseases from Cluj-Napoca, Romania, was decided.
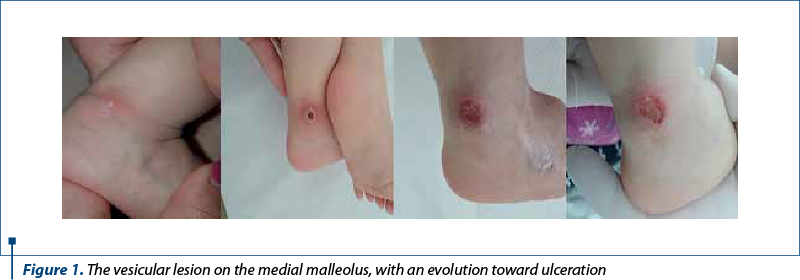
Upon admission, the girl presented influenced clinical status, sunken eyes, dry mouth and tongue, fever, cutaneous pallor, ulcerative lesion on the medial malleolus, oral aphthae, pharyngitis and bilateral cervical lymphadenopathy.
The initial laboratory studies revealed leukocytosis (29,090/mm3) with neutrophilia (20,710/mm3), normochromic, normocytic anemia (hemoglobin level 9.5 g/dl, MCV 80.1 fl, MCH 26.7 pg), thrombocytosis (490,000 platelets/mm3), a high level of CRP (9.66 mg/dl) and D-dimers (2.99), and a normal level of procalcitonin. The peripheral blood smear was without modifications. Cultures of blood, nasopharyngeal swab, urine and fecal samples were negative for bacteria or fungi. Also, the culture from the malleolar ulcerative lesion was negative. Other laboratory findings were within normal ranges. Antibiotic therapy with meropenem (60 mg/kg/day) and clindamycin (30 mg/kg/day) was initiated with antipyretics and hydrocortisone. The antibiotics were chosen considering the patient’s allergy to ceftriaxone and metronidazole. Despite the treatment, the fever and the inflammatory syndrome with leukocytosis, neutrophilia, high CRP and D-dimers persisted. The girl also presented a new vesicular lesion on the legs and arms. Vancomycin (40 mg/kg/day) and fluconazole (6 mg/kg/d) were added to the treatment.
Vasculitis and chronic granulomatous disease were taken into consideration. The immunological profile of the patient was unremarkable (normal level of IgG and IgA, normal level of rheumatoid factor, ANA and pANCA antibodies and normal level of C3 and C4). The thoracic radiography was without modifications. The abdominal ultrasound revealed two intrasplenic, hypoechogenic and avascular lesions, a first one of 20 mm, in the medial subcapsular area of the inferior pole, and another one of 8 mm in the hilar area, suggestive of splenic abscesses (Figure 2).
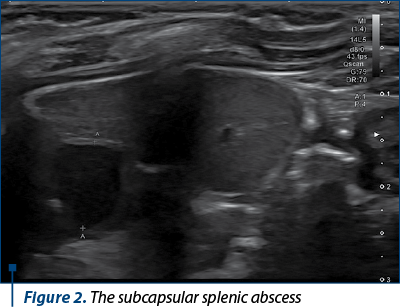
Endocarditis was ruled out, considering the patient’s clinical status and the possibility of association of endocarditis with splenic abscesses.
Based on this evidence, a splenic abscess secondary to hematological dissemination from the malleolar lesion was considered. For further investigations, the girl was transferred to our medical service.
At admission, she presented good clinical status, growth retardation (weight below the first percentile and length below the 10th percentile), pallor, ulcerative lesion on the medial malleolus, pharyngitis and bilateral cervical lymphadenopathies.
The laboratory tests revealed: leukocytosis (26,970/mm3) with neutrophilia (10,910/mm3), normochromic, normocytic anemia (hemoglobin level 10.7 g/dl, MCV 78.3 fl, MCH 25.4 pg, thrombocytosis (927,000 platelets/mm3), elevated ESR (33 mm/h) and normal CRP. Hypercholesterolemia and hypertriglyceridemia were found.
The antibiotic therapy with meropenem, vancomycin and clindamycin was continued in our clinic. Under the therapy, the splenic lesions were decreased, which confirmed the lesions’ infective origin. After 14 days of therapy, the hilar lesion was absent, and the subcapsular lesion had a diameter of 10 cm.
The level of IgA, IgM and IgG, lymphocytic subpopulations and genetic tests were performed for the suspicion of immune deficiency. Only a transient low level of IgA was found.
Her evolution was marked by dysbiosis with diarrhea and rash on the 46th day of therapy secondary to prolonged antibiotic therapy and superficial venous phlebitis on the left hand.
After 47 days of antibiotic therapy, only the subcapsular lesion was observed, measuring 3 cm, with a hyperechoic halo of 1.5 mm. Another four weeks later, the lesions were unremarkable.
Discussion
Splenic abscess is a rare entity, with an incidence of 0.4-0.7% in studies, based on autopsy, and with about 600 cases described until 2004(2,3). Most studies on case series with splenic abscesses have a maximum of 20 patients reported(8). The incidence is low because the spleen, part of the reticuloendothelial system, can fight against pathogens.
The abscess is usually a diagnosis by exclusion or an incidental finding. Splenic abscess represents a septic collection, which occurs as metastatic infection from other parts of the body or by direct extension from neighboring infections(1). Most regularly, the splenic abscess is a complication of infective endocarditis in about 10% of the cases. Urinary infections, otitis media, pneumonia, or intraabdominal infections might get complicated by splenic abscesses(5,9-11).
The abscesses usually occur in the immunosuppressed population, but they can also occur in immunocompetent children due to the dissemination of an infective agent to a preexisting splenic condition. One study reported a splenic abscess secondary to umbilical vein catheterization. In developing countries, the splenic abscess is found in typhoid fever. The most important etiologies and risk factors for splenic abscess are found in Table 1(1,4,6,12).

Organisms commonly associated with splenic abscesses are aerobe and anaerobe bacteria, but various rare organisms, fungi or parasites might also be involved. Monomicrobial growth is found on cultures in more than 50% of the cases. Enteric Gram-negative bacteria are found in two-thirds of the cases. Gram-positive cocci or anaerobes might also be involved. However, in 50% of the cases, cultures from the abscess or the hemoculture are negative, and the etiological agent is not found(1,4-6,10,11).
Recently, aseptic abscesses have also been described, particularly in patients with inflammatory bowel disease. The patients with aseptic abscesses present with fever and respond to corticosteroids but not to antibiotics. This was not the case in our girl, as long as she responded very well to antibiotics.
Although splenic abscesses were described more often in males older than 10 years, in our case, a girl of only 14 months old was affected(8). In the literature, only one case of an infant with a splenic abscess secondary to an umbilical vein catheter was reported. The other case reports are about older children(2,8,9).
A history of trauma and a modified spleen structure were excluded. The follow-up echography revealed the absence of the abscesses and a normal echo-structured echogenic spleen with normal Doppler flux. There was no evidence of malignancies, and immunodeficiencies were ruled out based on the immunological tests. Sickle cell anemia was not considered, although mild anemia was present, because it was transient during the infection, the peripheral blood smear was unmodified, and the incidence in our country is known as minimal. The predisposing factors are not always found (53% of the patients have underlying conditions)(8). Therefore, an abscess secondary to hematogenous spread from the skin lesion was considered, although the culture from the lesion and blood cultures were negatives. However, it is known that, in about 50% of the cases, blood cultures and even the cultures from the abscess can be aseptic(10). Hematogenous dissemination from a skin lesion to the spleen in an infant was not often described in literature. Skin lesions associated with splenic abscesses most often described until now were in the context of cat-scratch disease, brucellosis, melioidosis or aseptic splenic abscesses associated with pyoderma gangrenosum or neutrophilic dermatosis in Sweet syndrome(14-19). These diseases were clinically excluded.
The clinical presentation is usually insidious, vague and unspecific, as in our case. Patients with splenic abscess might present recurrent or persistent fever despite antibiotics, left upper quadrant abdominal pain, pleuritic chest pain (pleural effusion), left shoulder pain (diaphragmatic irritation), abdominal distention, tender mass on the left hypochondrium or splenomegaly. The triad of fever, left upper quadrant tenderness/abdominal pain and leukocytosis is found in one-third of the cases(4,5). On average, fever appears eight days before the diagnosis was established, it is persistent despite the treatment with antipyretics and hardly responds to the antibiotic treatment. It is found in more than 90% of patients. Generalized or localized abdominal pain and left quadrant tenderness are also frequent, reported in about 70% of the cases(1,9,10,20). Splenomegaly occurs only in one-third of the cases(20). Less frequently, splenic friction rubs, hepatomegaly or ascites are found. Our patient presented only a fever hardly responsive to antipyretics, without abdominal pain, although it is difficult to say that abdominal pain was absent considering the child’s age.
Laboratory tests reveal inflammatory syndrome. Leukocytosis is found in more than 80% of the cases, with a mean range of 19,500/mm3. It is usually associated with neutrophilia, high levels of CRP (mean range 13.5 mg/dl) and with ESR (mean range 74 mm/h)(10). In our case, the patient presented a level of leukocytes above the range, while CRP and ESR were below the range. In evolution, leukocytosis persisted more in comparison to CRP and ESR. Additionally, high levels of D-dimers, moderate anemia and thrombocytosis were found. Procalcitonin was within normal values. The blood and lesion cultures were negative, but it is well known in the literature that the cultures might be negative in 50% of the cases. Also, cultures from the abscess might be negative in about 50% of the cases(10).
The radiographs are sensitive but not specific in splenic abscesses, with abnormalities detected in more than 50% of the cases, such as pleural effusion, basilar infiltrates, elevated hemidiaphragm or shift of viscera. They were absent in our case.
The diagnosis was established based on the ultrasound (US) examination and the clinical and imagistic evolution under antibiotic therapy. Both CT and US have a sensitivity of around 90%. The US is often chosen because of its cost-effectiveness, availability and portability, and because it doesn’t require sedation(12).
The abscesses can be solitary, multiple or multiloculated, and on US examination they are represented as hypoechoic or anechoic areas of varying sizes, with regular areas of echodensity or gas pattern within the lesion. The spleen might be enlarged. On color Doppler, they are typically avascular, and on CT scan they are found as hypodense areas; on magnetic resonance imaging (MRI), the lesions have low signal intensity on T1 and high signal on T2 sequences. Solidary abscesses are more frequent in adults, while most children present multiple abscesses. In patients with endocarditis and associated septic emboli, they are wedge-shaped. The fungal microabscesses are hypodense, ring-enhancing and smaller than 2 cm(16).
In our patient, two hypoechogenic lesions smaller or equal to 2 cm were present. Multiple abscesses are associated with silent evolution and higher mortality, but our case is not an example in this direction(10).
The splenic abscess might complicate by perforation into the peritoneum in 6.6% of the cases, which can be life-threatening. Rupture into adjacent organs, such as the gastrointestinal tract, the pleural space or the lung parenchyma, can also occur(20).
Splenic abscesses require prompt treatment, considering the high mortality in untreated lesions. Splenectomy is the gold-standard therapy. There is no consensus on the treatment, especially in children, but intravenous antibiotic therapy (27-66 days), percutaneous drainage, open drainage, and partial or total splenectomy were all reported. No mortality difference was reported between the treatment of splenectomy, drainage, aspiration and intravenous antibiotic therapy alone. However, conservative treatments are preferred because they avoid OPSI with capsulated bacteria and intraerythrocytic parasites(21).
Percutaneous aspiration of splenic abscesses has the advantage of lower morbidity and mortality and allows the spleen’s preservation. It is a reliable technique for smaller, solitary or unilocular abscesses. Percutaneous drainage might be useful in unstable patients or in those with an unacceptably high surgical risk. If percutaneous aspiration or drainage fails, surgery is mandatory.
Antibiotic therapy should be with a broad spectrum, covering both Gram-positive and Gram-negative aerobes and anaerobes. In most cases, antibiotic therapy includes a third- or fourth-generation of cephalosporins or carbapenems, plus aminoglycosides, vancomycin and metronidazole. After culture results are obtained, antibiotic therapy can be narrowed(9). Usually, antibiotic therapy alone may be insufficient(20).
Our patient was allergic to a third-generation cephalosporin and metronidazole. In this perspective, our antibiotic therapy (meropenem, clindamycin and vancomycin) covered a broad spectrum of pathogens, with a good penetrance in the abscess fluid, especially clindamycin. We have also associated fluconazole, considering the clinical evolution and the echographic aspect. The antibiotic therapy duration was within the reported limits (47 days). Although antibiotic therapy is a conservative treatment, avoiding splenectomy and its complications, this treatment might have its disadvantages.
Prolonged antibiotic therapy is known to lead to adverse events (AE). The most common are diarrhea, rash, allergic reactions, venous-catheter-related complications, candidiasis, neutropenia, renal insufficiency and hepatotoxicity. In a cohort evaluating AE of prolonged antibiotic therapy, diarrhea was present in 29.1% of the patients, rash in 8%, nausea, vomiting and abdominal pain in 7%, allergy in 4.5% of the patients and catheter-related events (catheter occlusion, displacement or infection) in 13% of the patients. In the same cohort, one distinct adverse event was present in 31.9% of the patients, while in 7.5% three or more AE were present(22,23).
In our patient, three adverse events have appeared: diarrhea, superficial vein thrombosis related to catheterism, rash and antibiotic allergy. These events occur during therapy, at a median of around 20 days after starting therapy. Superficial vein thrombosis appeared after 21 days of therapy and diarrhea appeared after 24 days. The patient developed a rash at the end of the therapy, 47 days after initiating therapy, related to meropenem. She was allergic to ceftriaxone and metronidazole at the first administration. Despite the adverse events, antibiotic therapy alone was efficient in this case, with the resolution of the lesions.
Conclusions
Splenic abscesses are rare in children, being most frequent in immunosuppressed patients or in patients with endocarditis or systemic embolization. Their diagnosis is a challenge. The history, the physical exam and the laboratory tests are not sufficiently reliable to make this diagnosis. The diagnosis depends on the imaging studies. However, they might be present in children without underlying conditions such as a metastatic site from a skin infection. The cultures are not always positive. Antibiotic therapy only might be effective, avoiding splenectomy and its complications. Prolonged antibiotic therapy has also transient adverse effects.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
-
Tung C-C, Chen F-C, Lo C-J. Splenic abscess: an easily overlooked disease? Am Surg. 2006;72(4):322–5.
-
Aslam A, Ahmed Shatla ES, Imanullah S, Bedair EMA. Splenic Abscess: A Rare Complication of the UVC in Newborn. Case Rep Pediatr. 2014;903421.
-
Carbonell AM, Kercher KW, Matthews BD, Joels CS, Sing RF, Heniford BT. Laparoscopic Splenectomy for Splenic Abscess. Surg Laparosc Endosc Percutan Tech. 2004;289–91.
-
Bona R. Evaluation of splenomegaly and other splenic disorders in adults [Internet]. UptoDate. 2022 [cited 2022 Aug 14]. Available from: https://www.uptodate.com/contents/evaluation-of-splenomegaly-and-other-splenic-disorders-in-adults?sectionName=Abscess and infarction&search=splenic abscess&topicRef=2135&anchor=H741989576&source=see_link#H14392127
-
Lotfollahzadeh S, Mathew G, Zemaitis MR. Splenic Abscess. Vol. StatPearls[Internet]. Treasure Island (FL): StatPearls Publishing; 2022.
-
Kamaya A, Weinstein S, Desser TS. Multiple Lesions of the Spleen: Differential Diagnosis of Cystic and Solid Lesions. Semin Ultrasound, CT MRI. 2006;27(5):389–403.
-
Sinwar PD. Overwhelming post splenectomy infection syndrome – review study. Int J Surg. 2014;12(12):1314–6.
-
Faruque AV, Qazi SH, Arshad M, Anwar N. Isolated splenic abscess in children, role of splenic preservation. Pediatr Surg Int. 2013;29(8):787–90.
-
Kadian Y, Saroha V, Jindal N, Rattan K. Splenic abscess in children: A report of three patients. African J Paediatr Surg. 2009;6(2):106–9.
-
Iñiguez A, Butte JM, Zuñiga JM, Torres J, Llanos O. Splenic abscesses. Report of seven cases. Rev Med Chil. 2008;136(1):38–43.
-
Gupta S, Hastir A, Mathur R, Singh O, Shukla S. Splenic abscesses: Reports of two cases with review of the literature. Ann Trop Med Public Heal. 2012;5(3):273–9.
-
Chang K-C, Chuah S-K, Changchien C-S, Tsai T-L, Lu S-N, Chiu Y-C, et al. Clinical characteristics and prognostic factors of splenic abscess: a review of 67 cases in a single medical center of Taiwan. World J Gastroenterol. 2006;12(3):460–4.
-
Pangonis S, Patamasucon P, Fitzpatrick E. Pneumococcal Sepsis Complicated by Splenic Abscesses and Purpura Fulminans in a 15-Month-Old Child. J Investig Med High Impact Case Reports. 2016;4(1):1–4.
-
Pavone P, Gulizia C, D’Amico S, Grassi P, La Rosa G, Leotta R, et al. Splenic abscesses in childhood brucellosis: a case-based review. Eur Rev Med Pharmacol Sci. 2022;26(13):4863–71.
-
Mohan A, Manan K, Tan LS, Tan YC, Chin ST, Ahmad R, et al. Detection of spleen abscesses facilitates diagnosis of melioidosis in Malaysian children. Int J Infect Dis. 2020;98:59–66.
-
Sakulchit T, Ngu L, Chor YK, Ong GY. Utilization of Point-of-Care Ultrasound to Detect Splenic Microabscesses in Pediatric Melioidosis. Cureus. 2021;13(3):e13760.
-
Gkamprela E, Papadimitropoulos V, Papadopoulos N, Deutsch M. Multifocal splenic abscesses in immunocompetent adult due to cat-scratch disease. Hippokratia. 2016;20(4):306–8.
-
Brahimi N, Maubec E, Boccara O, Marinho E, Valeyrie-Allanore L, Lecaille C, et al. Pyoderma gangrenosum with aseptic spleen abscess. Ann Dermatol Venereol. 2009;136(1):46–9.
-
Klinger S, Mathis N, Jackson S. Bullous Sweet syndrome associated with an aseptic splenic abscess. Cutis. 2009;84(5):255–8.
-
Mustafa M, Menon J, Muniandy R, Rahman M, Tan T. Splenic Abscess: Etiology, clinical spectrum and Therapy. J Pharm. 2015;5:37–41.
-
Matsubayashi T, Matsubayashi R, Saito I, Tobayama S, Machida H. Splenic abscess in an infant caused by Streptococcus intermedius. J Infect Chemother. 2007;13(6):423–5.
-
Murphy JL, Fenn N, Pyle L, Heizer H, Hughes S, Nomura Y, et al. Adverse Events in Pediatric Patients Receiving Long-term Oral and Intravenous Antibiotics. Hosp Pediatr. 2016;6(6):330–8.
-
Olson SC, Smith S, Weissman SJ, Kronman MP. Adverse Events in Pediatric Patients Receiving Long-Term Outpatient Antimicrobials. J Pediatric Infect Dis Soc. 2015;4(2):119–25. a