Advancements in ultrasound technology and the integration of cell-free DNA testing into standard prenatal screening services have made it more feasible to identify fetal sex at an early stage. Fetal sex determination through sonography relies on different factors, depending on the gestational stage. In the late second trimester, it is determined by directly visualizing the external genitalia. However, in the late first and early second trimesters, it primarily relies on the direction of the genital tubercle, known as the sagittal sign. Other sonographic landmarks, like the fetal scrotum, midline raphe of the penis, labial lines, uterus, descended testis, and the direction or origin of the fetal micturition jet in males, can aid in the fetal sex determination accuracy. However, malformed external genitalia can lead to inaccurate results. This article focuses on the sonographic determination of fetal sex during the late first trimester, early second trimester and late second trimester, utilizing both transvaginal and transabdominal ultrasound techniques. Additionally, we explore various elements that have contributed to the precision of fetal sex determination. While ultrasound examination of the external genitalia is typically sufficient, there are certain situations where it becomes crucial to identify the internal genitalia for precise prenatal diagnosis and thorough counseling. This paper aims at reviewing these situations in which the evaluation of the external genital organs is necessary, as well as the ways in which ultrasound can be used in the diagnosis of genital malformations.
Advancements in ultrasound assessment of fetal genitalia for accurate diagnosis and comprehensive counseling
Progrese în evaluarea ecografică a organelor genitale fetale pentru un diagnostic precis şi consiliere corespunzătoare
First published: 27 noiembrie 2023
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.42.4.2023.8994
Abstract
Rezumat
Progresele în ecografie şi integrarea testelor neinvazive prenatale în standardul screeningului prenatal au determinat fezabilitatea identificării sexului fetal în stadiul incipient. Determinarea sexului fetal prin ecografie se bazează pe diferiţi factori, în funcţie de vârsta de gestaţie. La sfârşitul trimestrului al doilea, se determină prin vizualizarea directă a organelor genitale externe. Cu toate acestea, la sfârşitul primului trimestru şi începutul celui de-al doilea trimestru, se bazează în primul rând pe orientarea tuberculului genital. Alte repere ecografice, cum ar fi scrotul fetal, linia mediană a penisului, liniile labiale, uterul, testiculul coborât şi direcţia sau originea jetului de micţiune fetal la sexul masculin, pot ajuta la acurateţea determinării sexului fetal. Totuşi, organele genitale externe malformate pot conduce la rezultate incorecte. Acest articol se concentrează pe determinarea ecografică a sexului fetal la finalul primului trimestru, începutul celui de-al doilea trimestru, precum şi la finalul acestuia, utilizând atât tehnici ecografice transvaginale, cât şi transabdominale. În plus, explorăm diversele elemente care au contribuit la precizia determinării sexului fetal. Deşi examinarea ecografică a organelor genitale externe este de obicei suficientă, există anumite situaţii în care devine crucială identificarea organelor genitale externe, atât pentru un diagnostic prenatal precis, cât şi pentru o consiliere amănunţită. Lucrarea de faţă îşi propune să detalieze aceste situaţii în care este necesară evaluarea organelor genitale externe, precum şi modalităţile prin care ecografia poate fi utilizată în diagnosticul malformaţiilor genitale.
Introduction
In the context of several genetic disorders, ultrasound prediction of fetal gender can be of real utility(1). The first indications for ultrasound evaluation of fetal gender were pregnancies at risk of sex and X-linked pathologies. Currently, the fetal gender assessment is commonly done to satisfy the curiosity of the parents(2). Determining fetal sex has several clinical implications. For example, it can dictate the necessity of invasive testing in the case of X-linked conditions such as Duchenne muscular dystrophy and androgen insensitivity syndrome, or adrenoleukodystrophy(3,4). For male fetuses at risk of hemophilia, fetal gender assessment can be very useful in deciding the obstetrical management. Moreover, fetal sex evaluation can raise suspicion in the case of congenital adrenal hyperplasia which can facilitate early administration of steroids to the mother in order to improve outcomes in female babies(3).
Although fetal sex evaluation is usually conducted in the second or third trimester, it is now possible due to technological advancements to assess fetal sex in the first trimester(2). Several methods have been proposed and studied to predict fetal sex in the first trimester: the sagittal sign, the anogenital distance, three-dimensional (3D) ultrasound, measuring the angle between genital tubercle and lumbosacral skin surface etc.(5-7) In the first trimester, the efficacy of these methods is lower than in the second or third trimester ultrasound evaluation, with a high probability of false-negative results(8). Nowadays, the early identification of anomalies allows a lower rate of complications related to pregnancy termination(9).
Ultrasound assessment of fetal gender in the first trimester
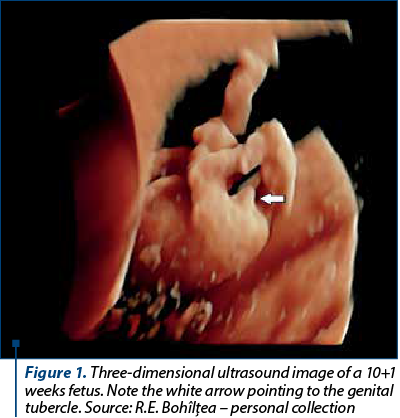
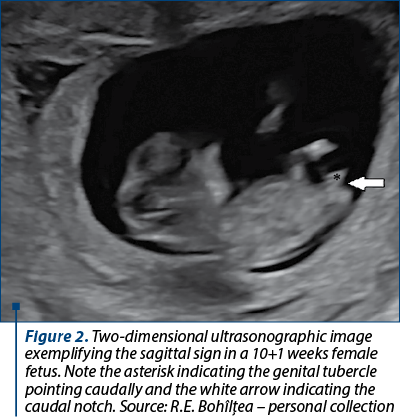
In the second and third trimesters, the visualization of the penis and scrotum suggests a male fetus, while the visualization of the labia minora and majora suggests a female fetus. Up until 14 gestational weeks, the difference in size between the penis and the clitoris is insignificant(10). Up until the 10th week of pregnancy, there is no difference between male and female external genitalia (Figure 1). After 12 weeks, changes in the urogenital sinus leads to the differentiation of external genital(5). One of the first ultrasound markers in predicting fetal gender in the first trimester was the direction in which the genital tubercle points: cranially for boys and caudally for girls(11) (Figure 2). Another ultrasound method is represented by the “dome sign’’, where the fetal scrotum appears as a non-septated dome-shaped structure at the base of the fetal penis. An echogenic line at the base of the penis is suggestive of the penile raphe and can help in diagnosing a male fetus. Visualization of two or four parallel lines is suggestive for both labia minora and majora, and it can help in diagnosing a female fetus(11).
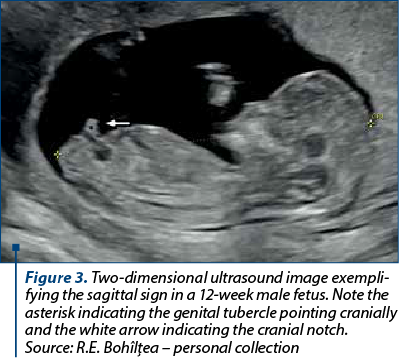
The sagittal sign represents the visualization of a cranial notch in male fetuses or a caudal notch in female fetuses(12) (Figure 3). This sign was evaluated by Mazza et al. in 2002. For a biparietal diameter (BPD) between 21 and 23 mm, the diagnosis of fetal sex was always correct(13). In another study, starting from a BPD of 22 mm, the fetal gender was correctly assigned in 99% to 100% of cases, the authors concluding that fetal sex prediction should not be done below this threshold(7).
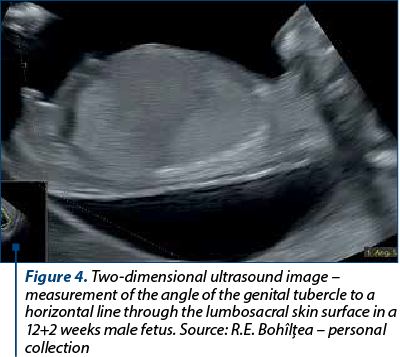
Efrat et al. evaluated another method of predicting fetal gender in the first trimester. Using a midsagittal plane, the angle between a horizontal line through the lumbosacral skin surface and the genital tubercle was determined (Figure 4). The fetus was assigned as male if the angle was greater than 30º and as a female if the angle was less than 30º. The fetal gender evaluation was accurate in 92.3% of cases, the accuracy increasing with gestational age from 70.3% at 11 weeks to 100% at 13 weeks. Therefore, the authors concluded that the decision for invasive testing in X-linked conditions should not be made before 12 weeks(5). Chelli et al. also used an angle measurement to assess fetal gender: for an angle greater than 30º, male sex was assigned, and for an angle less than 10º, female sex was assigned. The diagnosis was correct in 85.7% of cases(14).
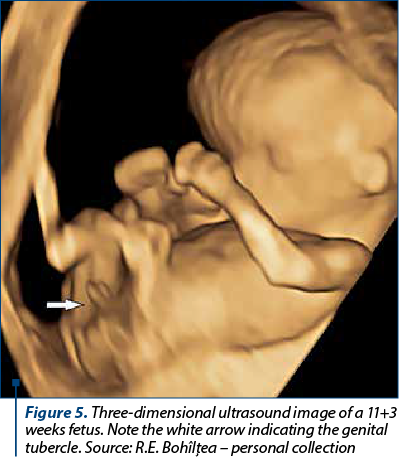
Bogers et al. evaluated the efficacy of 3D ultrasound in predicting fetal sex in the first trimester. This technique had a modest efficacy, with an accurate prediction in 54.9% of cases(15). Better results were provided by Michailidis et al., where two examiners evaluated 3D ultrasound images to predict fetal sex, with a correct prediction in 85.3% of cases(6) (Figure 5).
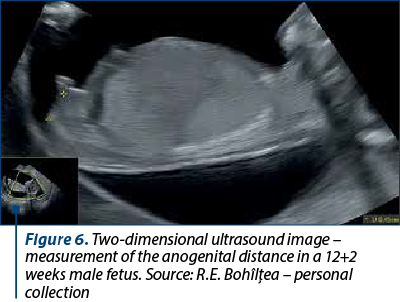
Anogenital distance as a method of predicting fetal sex was first evaluated by Arfi et al., who, using a cut-off of 4.8 mm, correctly identified 87% of males and 89% of females(16). In another study, by Sipahi et al., genders were demonstrated accurately for males in 76.7% of cases and for females in 97.1% of cases, using the same technique(17) (Figure 6).
Ultrasound evaluation of fetal gender in the second and third trimesters
Unlike the first trimester, the determination of the fetal sex in the second and third trimesters is based on the direct visualization of the external genital organs represented by the penis and scrotum in males, respectively the labia minora and labia majora in females. The accuracy of determining the fetal sex after the first trimester varies between 92% and 100% of cases, with a better accuracy in the case of the male sex supported by certain studies and with a better precision in the female sex supported by other studies(8).
In some cases, the simple evaluation of external genitalia can be insufficient and can require additional evaluation of other structures such as the fetal scrotum, testicular descent, dimensions of the penis, uterus characteristics in girls, and observing micturition in boys(8). Some of the least studied fetal structures are the fetal scrotum and its contents. The testicular descent does not take place until 24 weeks of pregnancy, which results in not evaluating testicular descent during the second trimester ultrasound. In addition, there are no international recommendations concerning the ultrasound examination of fetal genitalia(18). Ultrasonography can be useful in the prenatal diagnosis of several conditions such as cryptorchidism, inguinoscrotal hernia, intrauterine testicular torsion and meconium periorchitis(19-23).
Ultrasonographic evaluation of the scrotum and its content is feasible in 100% of cases between 34 and 36 weeks of gestation, the efficacy decreasing to 71.4% at 40 weeks due to reduced amniotic fluid and due to unfavorable fetal decubitus(24). Ultrasound can evaluate testicle descend, structural anomalies and scrotal masses. Several studies evaluated testicle descend and concluded that the descend is present in 5% of cases between 24 and 25 weeks, and it is present in 97% of cases between 32 and 34 weeks, the incidence of cryptorchidism being 3%(20,24,25). Structural anomalies involve the number, appearance or position abnormalities of the scrotum or testicles. Some examples of structural anomalies include scrotoschisis, bifid scrotum, or penoscrotal anomalies(26-28). Scrotal masses include testicular or paratesticular tumors such as rhabdomyosarcomas, lymphomas, teratomas, hemangiomas and metastases. These masses can be characterized by a solid or cystic aspect, the presence of calcifications, a positive Doppler signal, or the presence of peristalsis(29-31).
Intrauterine testicular torsion (IUTT) is a condition with unknown etiology which is usually diagnosed between 34 and 38 gestational weeks, or it can occur during delivery. Ultrasound evaluation shows a negative Doppler signal and a heterogenous testicle with hypoechoic areas that increased its size(32-34). Other ultrasound findings include contralateral hydrocele, lateralization of the scrotal septum toward the affected area, and the occurrence of hemorrhagic fluid at the level of the tunica vaginalis(35). Melcer et al. proposed an algorithm for managing IUTT. Bilateral IUTT after 34 gestational weeks requires emergency delivery in order to save at least one of the testicles. In the case of bilateral IUTT under 34 weeks or unilateral IUTT, the therapeutic management is expectant, the birth taking place according to the obstetrical indications. The exception is represented by the situation in which there is a unilateral IUTT with an onset of a maximum of 24 hours diagnosed during successive ultrasound evaluations. This situation requires emergency delivery(19).
Regarding the ultrasound evaluation of the penis, there is no consensus regarding the used methods, as well as regarding the normal upper and lower limits. In addition to the classic measurements of length, width and diameter, structures such as the glans, corpora cavernosa and corpora spongiosa can now be assessed using ultrasound(18). The most frequently used penile length measuring method involves measuring the distance between the tip of the penis and the edge of the scrotum, this distance being called outer penile length (OPL)(36). Besides OPL, other authors used different methods to determine the length of the penis. They used the distance between the tip of the penis and the proximal edge of the cavernous(37), or spongiosus corpus(38), or the distance between the tip of the penis and the pubic symphysis(36).
Using the data from clinical studies, the average length of the penis between 19 and 22 weeks should be between 7 and 10 mm, a length below 4 mm suggesting the diagnosis of micropenis. There are no cut-off values for the diagnosis of macropenis(18). Some authors created reference tables for fetal penis dimensions(39). When we are dealing with a macropenis, it is most often associated with megalourethra. In this situation, a careful evaluation of the urinary system and amniocentesis are mandatory(40). In case of an isolated macropenis, it can raise the suspicion of congenital adrenal hyperplasia. In this situation, amniocentesis for karyotype and analysis of steroid metabolites from the amniotic fluid are recommended(41). The diagnostic approach in case of a micropenis requires a detailed ultrasound scan focused on the genitourinary system that will later guide the choice of a genetic test in order to establish whether the micropenis is in the context of a chromosomal or non-chromosomal abnormality(41). Visualization of fetal micturition can help in the diagnosis of fetal hypospadias(8).
The small size of the ovaries can prevent their visualization by prenatal ultrasonographic evaluation. Therefore, these structures become visible in case of some pathologies that cause their volume to increase. For example, fetal ovarian cysts can be detected prenatally using ultrasound(42). The fetal uterus has a linear growth and can be visualized sonographically starting at 19 weeks(43). However, in 20% of cases, the fetal uterus cannot be detected by 2D ultrasound. With regard to 3D ultrasound, it managed to visualize the fetal uterus in less than 50% of cases between 20 and 22 weeks, with the detection efficiency increasing to 80% between 30 and 32 weeks(44). In situations where the uterus is not visible, an indirect sign can be used, involving the evaluation of the distance between the urinary bladder and the rectum, which is greater in females than in males due to the interposition of the uterus between these organs. Also, the uterus determines a concave indentation at the level of the posterior wall of the urinary bladder. Using this method, Glanc et al. managed to correctly predict the female gender in 98.8% of cases and the male gender in 100% of cases(45).
Prenatal management of chromosomal sex and phenotypic sex discrepancies, or ambiguous genitalia
The incidence of contradictory results between the ultrasound prediction of the fetal sex and the noninvasive prenatal testing (NIPT) result is below 0.01%(46). Approximately one-third of these inconsistencies are due to errors such as the wrong interpretation of ultrasound images or the wrong labelling of samples with biological products(46). Although NIPT has a good accuracy in detecting trisomies 21, 13 and 18, it did not have the same efficacy in detecting sex chromosome aneuploidy, only one-third of these cases being confirmed by karyotyping(47,48). Repeated contradictory results between the ultrasound prediction of the fetal sex and NIPT require invasive prenatal testing, amniocentesis being the most appropriate method, since chorionic villus sampling has a high rate of confined placental mosaicism associated with sex chromosome anomalies(48,49).
Despite the fact that ultrasound has good accuracy, rare anomalies are still difficult to diagnose prenatally(50). When it comes to ambiguous genitalia, the ultrasound findings should be described in detail, with careful attention to female or male genitalia characteristics(42). Careful ultrasound evaluation of the fetal anatomy can highlight other anomalies or particular signs that can guide the diagnosis. Genetic counseling with anamnesis, including a detailed family history, will guide the type of genetic testing required. Conventional karyotype can bring a lot of information, but karyotype with FISH (fluorescence in situ hybridization) for SRY (sex reversal Y) and hormonal analysis of amniotic fluid may be necessary(51). When a specific syndrome is suspected, specific genetic tests, such as SNP (single nucleotide polymorphism) array and exome or whole genome sequencing, can help the clinician. Prenatal follow-up should include multiple ultrasound assessments in order to detect any fetal impairment, such as intrauterine growth restriction or oligohydramnios(42).
Conclusions
Beyond satisfying the parents’ curiosity, predicting fetal sex can become a necessity in certain situations. This can determine the indication of invasive prenatal testing in the case of X-linked diseases, it can determine the obstetric management of fetuses at high risk of hemophilia, and it can dictate the initiation of treatment in case of congenital adrenal hyperplasia. Although ultrasound evaluation in the first trimester is not as effective as in the second and third trimesters and considering the very good effectiveness of NIPT in detecting fetal sex, ultrasonographic prediction of fetal sex in the first trimester may remain an option in countries with limited financial resources. Fetal genital pathology is vast, with numerous etiologies and, despite its complexity, it is insufficiently studied, with a lack of guidelines to dictate the management of these high-risk pregnancies. Future studies should focus on a better understanding of these pathologies and on the establishment of prenatal diagnosis and management protocols of these complex conditions, thus helping clinicians provide adequate advice to parents and directing complicated cases to centers where postnatal reconstructive surgeries can be performed.
Conflict of interest: none declared.
financial support: none declared.
This work is permanently accessible online free of charge and published under the CC-BY licence.

Bibliografie
-
Gharekhanloo F. The ultrasound identification of fetal gender at the gestational age of 11-12 weeks. J Family Med Prim Care. 2018;7(1):210-12.
-
Kearin M, Pollard K, Garbett I. Accuracy of sonographic fetal gender determination: predictions made by sonographers during routine obstetric ultrasound scans. Australas J Ultrasound Med. 2014;17(3):125-30.
-
Lewis C, Hill M, Skirton H, Chitty LS. Non-invasive prenatal diagnosis for fetal sex determination: benefits and disadvantages from the service users’ perspective. Eur J Hum Genet. 2012;20(11):1127-33.
-
Ovidiu B, Marcu DR, Mischianu DLD, Poiană C, Diaconu CC, Bungau SG, Tit DM, Cumpănaş A, Bohîlţea R. The challenges of androgen insensitivity syndrome. Arch Med Sci. 2021;18(4):881-9.
-
Efrat Z, Akinfenwa OO, Nicolaides KH. First-trimester determination of fetal gender by ultrasound. Ultrasound Obstet Gynecol. 1999;13(5):305-7.
-
Michailidis GD, Papageorgiou P, Morris RW, Economides DL. The use of three-dimensional ultrasound for fetal gender determination in the first trimester. Br J Radiol. 2003;76(907):448-51.
-
Mazza V, Di Monte I, Pati M, Contu G, Ottolenghi C, Forabosco A, Volpe A. Sonographic biometrical range of external genitalia differentiation in the first trimester of pregnancy: analysis of 2593 cases. Prenat Diagn. 2004;24(9):677-84.
-
Odeh M, Granin V, Kais M, Ophir E, Bornstein J. Sonographic fetal sex determination. Obstet Gynecol Surv. 2009;64(1):50-7.
-
Munteanu O, Cîrstoiu MM, Filipoiu FM, Neamţu MN, Stavarache I, Georgescu TA, Bratu OG, Iorgulescu G, Bohîlţea RE. The etiopathogenic and morphological spectrum of anencephaly: a comprehensive review of literature. Rom J Morphol Embryol. 2020;61(2):335-43.
-
Colmant C, Morin-Surroca M, Fuchs F, Fernandez H, Senat MV. Non-invasive prenatal testing for fetal sex determination: is ultrasound still relevant? Eur J Obstet Gynecol Reprod Biol. 2013;171(2):197-204.
-
Bronshtein M, Rottem S, Yoffe N, Blumenfeld Z, Brandes JM. Early determination of fetal sex using transvaginal sonography: technique and pitfalls. J Clin Ultrasound. 1990;18(4):302-6.
-
Emerson DS, Felker RE, Brown DL. The sagittal sign. An early second trimester sonographic indicator of fetal gender. J Ultrasound Med. 1989;8(6):293-7.
-
Mazza V, Falcinelli C, Percesepe A, Paganelli S, Volpe A, Forabosco A. Non-invasive first trimester fetal gender assignment in pregnancies at risk for X-linked recessive diseases. Prenat Diagn. 2002;22(10):919-24.
-
Chelli D, Methni A, Dimassi K, Boudaya F, Sfar E, Zouaoui B, Chelli H, Chennoufi MB. Fetal sex assignment by first trimester ultrasound: a Tunisian experience. Prenat Diagn. 2009;29(12):1145-8.
-
Bogers H, Rifouna MS, Koning AHJ, Husen-Ebbinge M, Go ATJI, van der Spek PJ, Steegers-Theunissen RPM, Steegers EAP, Exalto N. Accuracy of fetal sex determination in the first trimester of pregnancy using 3D virtual reality ultrasound. J Clin Ultrasound. 2018;46(4):241-6.
-
Arfi A, Cohen J, Canlorbe G, Bendifallah S, Thomassin-Naggara I, Darai E, Benachi A, Arfi JS. First-trimester determination of fetal gender by ultrasound: measurement of the ano-genital distance. Eur J Obstet Gynecol Reprod Biol. 2016;203:177-81.
-
Sipahi M, Tokgöz VY, Alanya Tosun Ş. An appropriate way to predict fetal gender at first trimester: anogenital distance. J Matern Fetal Neonatal Med. 2019;32(12):2012-6.
-
Soto ÁL, González JLM, Martínez MV, Pérez RL, Rivero IM, Fernández ML, Izquierdo OG, Cendán JPM. Sonographic evaluation of fetal scrotum, testes and epididymis. Obstet Gynecol Sci. 2021;64(5):393-406.
-
Melcer Y, Mendlovic S, Klin B, Keidar R, Lysyy O, Herman A, Maymon R. Fetal diagnosis of testicular torsion: what shall we tell the parents? Prenat Diagn. 2015;35(2):167-73.
-
Zampieri N, Zamboni C, Ghidini A, Borruto F, Camoglio FS. Prenatal sonographic evaluation of male genitalia development. Minerva Ginecol. 2008;60(4):317-21.
-
Braga LH, Lorenzo AJ, Romao RLP. Canadian Urological Association-Pediatric Urologists of Canada (CUA-PUC) guideline for the diagnosis, management, and followup of cryptorchidism. Can Urol Assoc J. 2017;11(7):E251-E260.
-
Ronzoni S, Melamed N, Kingdom JC, Ryan G, Jaeggi E, Windrim RC. Prenatal diagnosis of inguinoscrotal hernia associated with bowel dilatation: a pathogenetic hypothesis. Prenat Diagn. 2015;35(11):1151-3.
-
Durmuş G, Boybeyi-Türer Ö, Gharibzadeh-Hizal M, Ekinci S, Kiper N. Meconium periorchitis: An incidentally diagnosed rare entity during inguinal herniorraphy. Turk J Pediatr. 2018;60(5):612-4.
-
Fait G, Yaron Y, Shenhar D, Gull I, Har-Toov J, Jaffa AJ, Wolman I. Sonographic detection of undescended testes in the third trimester. J Ultrasound Med. 2002;21(1):15-8; quiz 20.
-
Achiron R, Pinhas-Hamiel O, Zalel Y, Rotstein Z, Lipitz S. Development of fetal male gender: prenatal sonographic measurement of the scrotum and evaluation of testicular descent. Ultrasound Obstet Gynecol. 1998;11(4):242-5.
-
DeRoo SE, Lumpkins KM, El-Metwally DE. Scrotoschisis in a neonate with meconium peritonitis and periorchitis. J Neonatal Perinatal Med. 2016;9(1):107-12.
-
Inde Y, Terada Y, Ikegami E, Sekiguchi A, Nakai A, Takeshita T. Bifid scrotum and anocutaneous fistula associated with a perineal lipomatous tumor complicated by temporary bilateral cryptorchidism in utero mimicking ambiguous genitalia: 2-D/3-D fetal ultrasonography. J Obstet Gynaecol Res. 2014;40(3):843-8.
-
Fahmy MA, El Shennawy AA, Edress AM. Spectrum of penoscrotal positional anomalies in children. Int J Surg. 2014;12(9):983-8.
-
Suriawinata A, Talerman A, Vapnek JM, Unger P. Hemangioma of the testis: report of unusual occurrences of cavernous hemangioma in a fetus and capillary hemangioma in an older man. Ann Diagn Pathol. 2001;5(2):80-3.
-
Paladini D, Palmieri S, Morelli PM, Forleo F, Morra T, Salviati M, Zampella C, D’Angelo A, Martinelli P. Fetal inguinoscrotal hernia: prenatal ultrasound diagnosis and pathogenetic evaluation. Ultrasound Obstet Gynecol. 1996;7(2):145-6.
-
Stupak A, Krzyzanowski A, Semczuk-Sikora A, Dymanowska-Dyjak I, Geca T, Kondracka A, Kwasniewska A. Conservative management after prenatal ultrasound diagnosis of meconium periorchitis. J Med Ultrason. 2014;41(4):499-505.
-
van der Sluijs JW, den Hollander JC, Lequin MH, Nijman RM, Robben SG. Prenatal testicular torsion: diagnosis and natural course. An ultrasonographic study. Eur Radiol. 2004;14(2):250-5.
-
Al-Salem AH. Intra-uterine testicular torsion: early diagnosis and treatment. BJU Int. 1999;83(9):1023-5.
-
Arena F, Nicòtina PA, Romeo C, Zimbaro G, Arena S, Zuccarello B, Romeo G. Prenatal testicular torsion: ultrasonographic features, management and histopathological findings. Int J Urol. 2006;13(2):135-41.
-
Herman A, Schvimer M, Tovbin J, Sandbank J, Bukovski I, Strauss S. Antenatal sonographic diagnosis of testicular torsion. Ultrasound Obstet Gynecol. 2002;20(5):522-4.
-
Nemec SF, Nemec U, Weber M, Brugger PC, Bettelheim D, Rotmensch S, Krestan CR, Rimoin DL, Graham JM Jr, Prayer D. Penile biometry on prenatal magnetic resonance imaging. Ultrasound Obstet Gynecol. 2012;39(3):330-5.
-
Perlitz Y, Keselman L, Haddad S, Mukary M, Izhaki I, Ben-Ami M. Prenatal sonographic evaluation of the penile length. Prenat Diagn. 2011;31(13):1283-5.
-
Vuillard E, Chitrit Y, Dreux S, Elghoneimi A, Oury JF, Muller F. Sonographic measurement of corpus spongiosum in male fetuses. Prenat Diagn. 2011;31(12):1160-3.
-
López Soto Á, Velasco Martínez M, Meseguer González JL, López Pérez R. Third trimester ultrasound. A long-standing debate. Taiwan J Obstet Gynecol. 2021;60(3):401-4.
-
Anh DD, Nguyen HT, Meagher S, Araujo Júnior E. Prenatal diagnosis of congenital megalourethra in the second trimester of pregnancy. J Ultrason. 2019;19(79):302-4.
-
Pajkrt E, Petersen OB, Chitty LS. Fetal genital anomalies: an aid to diagnosis. Prenat Diagn. 2008;28(5):389-98.
-
Mallmann MR, Gembruch U. Clinical spectrum of female genital malformations in prenatal diagnosis. Arch Gynecol Obstet. 2022;306(6):1847-62.
-
Soriano D, Lipitz S, Seidman DS, Maymon R, Mashiach S, Achiron R. Development of the fetal uterus between 19 and 38 weeks of gestation: in-utero ultrasonographic measurements. Hum Reprod. 1999;14(1):215-8.
-
Jouannic JM, Rosenblatt J, Demaria F, Jacobs R, Aubry MC, Benifla JL. Contribution of three-dimensional volume contrast imaging to the sonographic assessment of the fetal uterus. Ultrasound Obstet Gynecol. 2005;26(5):567-70.
-
Glanc P, Umranikar S, Koff D, Tomlinson G, Chitayat D. Fetal sex assignment by sonographic evaluation of the pelvic organs in the second and third trimesters of pregnancy. J Ultrasound Med. 2007;26(5):563-9; quiz 570-1.
-
Dhamankar R, DiNonno W, Martin KA, Demko ZP, Gomez-Lobo V. Fetal Sex Results of noninvasive prenatal testing and differences with ultrasonography. Obstet Gynecol. 2020;135(5):1198-206.
-
Borth H, Teubert A, Glaubitz R, Knippenberg S, Kutur N, Winkler T, Eiben B. Analysis of cell-free DNA in a consecutive series of 13,607 routine cases for the detection of fetal chromosomal aneuploidies in a single center in Germany. Arch Gynecol Obstet. 2021;303(6):1407-14.
-
Lüthgens K, Grati FR, Sinzel M, Häbig K, Kagan KO. Confirmation rate of cell free DNA screening for sex chromosomal abnormalities according to the method of confirmatory testing. Prenat Diagn. 2021;41(10):1258-63.
-
Wang L, Meng Q, Tang X, Yin T, Zhang J, Yang S, Wang X, Wu H, Shi Q, Jenkins EC, Zhong N, Gu Y. Maternal mosaicism of sex chromosome causes discordant sex chromosomal aneuploidies associated with noninvasive prenatal testing. Taiwan J Obstet Gynecol. 2015;54(5):527-31.
-
Bohîlţea RE, Dumitru A, Vlădăreanu R, Pleş L, Georgescu TA, Petrescu IA, Munteanu O. Ultrasound pitfalls in a complex fetal cardiac malformation-case report of a new arteriovenous central communication. Diagnostics (Basel). 2021;11(12):2398.
-
Adam MP, Fechner PY, Ramsdell LA, Badaru A, Grady RE, Pagon RA, McCauley E, Cheng EY, Parisi MA, Shnorhavorian M. Ambiguous genitalia: what prenatal genetic testing is practical? Am J Med Genet A. 2012;158A(6):1337-43.
Articole din ediţiile anterioare
Ultrasound-based differential diagnosis of fetal abdominal wall defects in early pregnancy
Fetal abdominal wall defects (AWDs) encompass a wide range of congenital anomalies that involve the incomplete closure or disruption of the abdomin...
Performance of imaging exams in screening and diagnosis of breast cancer
Breast cancer holds great importance as a public health issue, threatening the physical and mental well-being of women worldwide. According to the ...
Body stalk anomaly – case report and review
Body stalk anomaly is a rare congenital defect with an incidence rate ranging from 1 in 14,000 to 1 in 31,000 pregnancies, according to large epide...
Prenatal diagnosis of micrognathia
Early detection of micrognathia during prenatal care is crucial to mitigate the associated risks and prevent unforeseen emergencies requiring inv...