Clear cell carcinoma of the ovary is a rare and aggressive subtype of ovarian cancer. It is often associated with endometriosis, a chronic condition characterized by the presence of endometrial-like tissue outside the uterus. Clear cell carcinoma arising within the context of endometriosis presents unique diagnostic challenges for pathologists. The coexistence of benign endometriotic tissue and malignant clear cell carcinoma in the same specimen can be confounding.
Clear cell carcinoma arising from endometriotic ovarian cyst
Carcinomul cu celule clare dezvoltat pe chist ovarian endometriozic
First published: 30 septembrie 2023
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.41.3.2023.8739
Abstract
Rezumat
Carcinomul cu celule clare este un subtip rar şi agresiv de cancer ovarian. Este adesea asociat cu endometrioza, o afecţiune cronică ce este caracterizată prin prezenţa de ţesut asemănător endometrului în afara uterului. Carcinomul cu celule clare care apare în contextul endometriozei prezintă provocări unice de diagnostic pentru patologi. Coexistenţa ţesutului endometriotic benign şi a carcinomului cu celule clare în acelaşi specimen poate induce confuzii.
Clear cell carcinoma (CCC) of the ovary is a unique and relatively uncommon histological subtype of epithelial ovarian cancer. A distinctive feature of CCC is its association with endometriosis, a benign gynecological condition where endometrial-like tissue is found outside the uterus. Understanding the development, clinical characteristics, diagnosis and treatment of CCC arising from endometriotic ovarian cysts is essential for improving patient care and outcomes.
Clear cell carcinoma arising within the context of endometriosis presents unique diagnostic challenges for pathologists. The coexistence of benign endometriotic tissue and malignant clear cell carcinoma in the same specimen can be confounding(1). This case report delves into the intricacies and challenges that pathologists encounter when tasked with distinguishing CCC from endometriotic cysts and other histological mimics.
The precise etiology of CCC originating from endometriotic ovarian cysts remains an area of active research. Several hypotheses have been proposed, including:
-
inflammation and oxidative stress – chronic inflammation in the context of endometriosis may induce genetic mutations and epigenetic changes, promoting the development of CCC;
-
hormonal and microenvironmental factors – altered hormonal and microenvironmental factors within endometriotic cysts may create a niche conducive to malignant transformation(2,3).
Case report
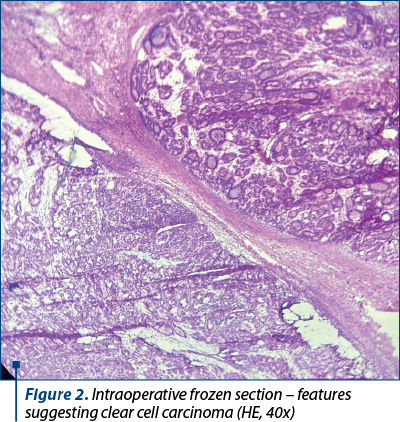
A 34-year-old patient presented to the Gynecology Clinic of the “Prof Dr. Panait Sîrbu” Clinical Hospital of Obstetrics and Gynecology, Bucharest, with pelvic-abdominal pain. The clinical examination identified a right ovary enlarged in volume, of approximately 9 cm in diameter. During the vaginal examination, a healthy cervix was observed. However, the uterus appeared enlarged. Other findings from the systemic examination did not provide relevant information. Consequently, the patient was prepared for an exploratory laparotomy. Intraoperatively, a tumor in the right adnexal region was identified. A sample of the right adnexal mass was obtained and sent for a frozen section analysis, which led to the diagnosis of clear cell carcinoma (Figures 1 and 2). Subsequently, the patient underwent a transabdominal hysterectomy with bilateral salpingo-oophorectomy.
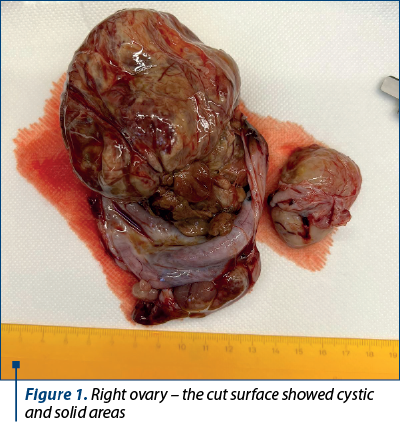
The right adnexal mass exhibited dimensions of 8.5×6.5×4.5 cm. On its external surface, nodular characteristics were observed. The cut surface revealed a combination of cystic brownish regions with solid yellowish areas. In contrast, the uterus and the left-sided adnexa exhibited no remarkable features.
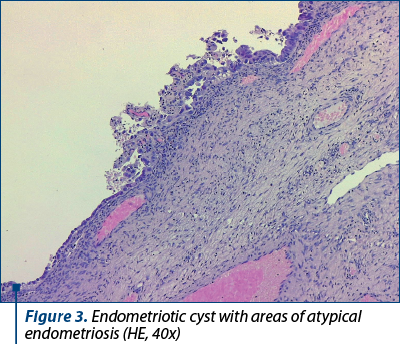
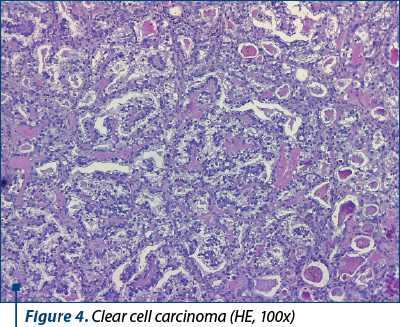
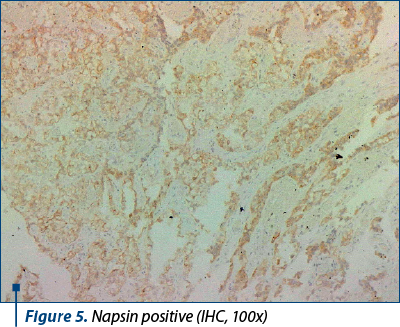
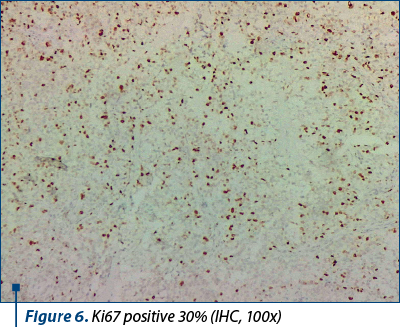
Within the right ovary, typical features of endometriosis were identified, characterized by the presence of endometrial glands and a dense stromal component. Clear cell carcinoma areas displayed cells arranged in solid sheets, tubulopapillary patterns and focal cribriform structures, separated by hyalinized homogeneous fibrovascular septae, all originating within an endometriotic cyst. Notably, hobnailing of the cells was evident. Immunohistochemistry was conducted to further characterize the tumor. The results indicated a wild-type p53 reaction, positivity for ER, napsin and EMA, and a lack of immunoreactivity for WT1. Based on the comprehensive evaluation, the diagnosis of clear cell carcinoma was conclusively confirmed (Figures 3-6).
Discussion
Clear cell carcinoma originating from endometriotic ovarian cysts often presents with subtle or nonspecific symptoms, making the early diagnosis challenging. The common clinical features include:
-
pelvic pain – patients may experience chronic pelvic pain, which is often misattributed to endometriosis;
-
ascites – the accumulation of ascitic fluid is a characteristic finding in advanced cases;
-
unilateral ovarian mass – the imaging studies may reveal a unilateral ovarian mass that could be misdiagnosed as a benign endometriotic cyst.
One of the primary diagnostic challenges in identifying CCC within endometriotic lesions is represented by the significant histological overlap between the two entities. Endometriosis often contains glands and stroma that bear a striking resemblance to the architectural features of CCC. These overlapping characteristics can include glandular structures, as both endometriosis and clear cell carcinoma may exhibit glandular formations and the presence of hobnail cells, which are characteristic of CCC, but these can occasionally be found in endometriotic cysts, further complicating the distinction.
To differentiate clear cell carcinoma from endometriosis, immunohistochemical staining can be employed. While immunostains for markers such as CK7, CK20, napsin, CD10 and HNF1B can be informative, their interpretation is not always straightforward, as both CCC and endometriosis can demonstrate overlapping staining patterns(4). Pathologists must carefully evaluate the staining results in the context of the entire histological picture.
Another diagnostic challenge is represented by the sampling of the specimen. Given the spatial heterogeneity of endometriosis and CCC within the same lesion, it is possible that a single biopsy may not fully capture the malignant component. Comprehensive sampling of the lesion, which often requires a detailed gross examination, is required.
Intraoperative frozen section analysis can be requested by surgeons to aid in decision-making during surgery. This further places responsibility on pathologists to provide a rapid and accurate diagnosis. However, CCC may not always present with definitive features in frozen sections, and the diagnosis may remain inconclusive until permanent sections are examined.
Recent advances in molecular and genetic profiling have shown promise in differentiating clear cell carcinoma from endometriosis. These approaches may include identifying specific genetic alterations or markers associated with malignancy. While not yet part of routine practice, molecular techniques hold potential for improving diagnostic accuracy in the future.
Recent research has shed light on the genetic alterations that may play a role in the development of CCC within ovarian endometriosis. Mutations in genes such as ARID1A and PIK3CA are frequently observed in clear cell carcinoma(5). Understanding these genetic changes is crucial for the development of targeted therapies and for identifying potential biomarkers for early detection. Distinguishing clear cell carcinoma within the context of endometriosis remains a complex task for pathologists due to the histological overlap and heterogeneity of these lesions. The accurate diagnosis of clear cell carcinoma is essential for determining the appropriate treatment strategy, as the management of endometriosis and CCC varies significantly. Therefore, pathologists must rely on a combination of histological assessment, immunohistochemical staining, molecular profiling, and clinical correlation to reach a definitive diagnosis. Further research and standardization of diagnostic criteria are needed to address these diagnostic challenges effectively.
Two distinct histological subtypes of ovarian cancer, clear cell carcinoma and endometrioid adenocarcinoma, are often associated with endometriosis and represent the main differential diagnosis.
Endometrioid adenocarcinoma is believed to arise from a similar pathway as clear cell carcinoma. It is postulated that endometriotic tissue undergoes malignant transformation due to genetic and epigenetic changes, similar to CCC. Mutations in PTEN, CTNNB1 and KRAS genes have been identified in endometrioid adenocarcinomas associated with endometriosis(6).
Endometrioid adenocarcinoma typically presents as an ovarian mass and may be associated with concurrent endometrial carcinoma. Histologically, it exhibits glandular formations resembling normal endometrial tissue, further suggesting a shared histogenic origin with endometriosis.
The dichotomy in the histogenesis of clear cell carcinoma and endometrioid adenocarcinoma within the context of endometriosis underscores the complex relationship between benign and malignant processes. Despite the shared association with endometriosis, the distinct genetic alterations and morphological characteristics of these two subtypes emphasize that their development is not uniform. The molecular and genetic pathways that drive their histogenesis continue to be the subject of active research.
The treatment of clear cell carcinoma arising from endometriotic ovarian cysts typically involves a multimodal approach(7):
-
surgery – optimal cytoreductive surgery is the cornerstone of treatment, aiming to remove as much tumor as possible;
-
chemotherapy – platinum-based chemotherapy regimens are commonly used as adjuvant therapy or in advanced cases;
-
targeted therapies – emerging targeted therapies are under investigation to improve outcomes, particularly for patients with recurrent disease.
The prognosis of clear cell carcinoma arising from endometriotic ovarian cysts is generally less favorable than for other ovarian cancer subtypes. The factors that impact prognosis include the stage at diagnosis – early-stage disease is associated with better outcomes, residual disease after surgery and with response to chemotherapy, the patients with platinum-sensitive disease having a more favorable prognosis.
Clear cell carcinoma arising from endometriotic ovarian cysts is a unique and challenging entity. Early diagnosis remains a formidable task due to the clinical and histological overlap with endometriosis. Individualized management strategies, including comprehensive surgical intervention and evolving targeted therapies, hold promise for improving patients’ outcomes. A better understanding of the etiology and pathogenesis of clear cell carcinoma in the context of endometriosis is essential for advancing our knowledge and for optimizing treatment approaches.
Conflict of interest: none declared.
financial support: none declared.
This work is permanently accessible online free of charge and published under the CC-BY licence.

Bibliografie
-
Wei JJ, William J, Bulun S. Endometriosis and ovarian cancer: a review of clinical, pathologic, and molecular aspects. Int J Gynecol Pathol. 2011;30(6):553-68.
-
Samartzis EP, Labidi-Galy SI, Moschetta M, Uccello M, Kalaitzopoulos DR, Perez-Fidalgo JA, Boussios S. Endometriosis-associated ovarian carcinomas: insights into pathogenesis, diagnostics, and therapeutic targets-a narrative review. Ann Transl Med. 2020;8(24):1712.
-
Kobayashi H, Yamada Y, Kawahara N, Ogawa K, Yoshimoto C. Integrating modern approaches to pathogenetic concepts of malignant transformation of endometriosis. Oncol Rep. 2019;41(3):1729-38.
-
Soslow RA. Histologic subtypes of ovarian carcinoma: An overview. Int J Gynecol Pathol. 2008;27(2):161–74.
-
De P, Dey N. Mutation-driven signals of ARID1A and PI3K pathways in ovarian carcinomas: alteration is an opportunity. Int J Mol Sci. 2019;20(22):5732.
-
Kajihara H, Yamada Y, Shigetomi H, Higashiura Y, Kobayashi H. The dichotomy in the histogenesis of endometriosis-associated ovarian cancer: Clear cell-type versus endometrioid-type adenocarcinoma. Int J Gynecol Pathol. 2012;31(4):304–12.
-
Takano M, Tsuda H, Sugiyama T. Clear cell carcinoma of the ovary: is there a role of histology-specific treatment? J Exp Clin Cancer Res. 2012;31(1):53.
Articole din ediţiile anterioare
Diagnosticul imagistic al endometriozei profund infiltrative
Endometrioza profund infiltrativă (EPI) reprezintă, prin definiţie, infiltrarea implanturilor endometriozice în profunzime mai mult de 5 mm de l...
IRM – avantaje şi limite în diagnosticul şi tratamentul endometriozei
Endometrioza este o boală ginecologică multifocală comună, care se manifestă în timpul anilor de reproducere, cauzând adesea dureri pelviene croni...
Managementul perioperator al unui pacient cu tumoră Krukenberg - studiu de caz
Tumora Krukenberg, cunoscută şi sub denumirea de carcinom mucocelular, se referă la o tumoră metastatică inelară rară a ovarului, localizarea prima...
Calitatea vieţii la pacientele cu endometrioză şi rezecţie colorectală laparoscopică
Obiectiv. Scopul acestei lucrări este de a evalua eficacitatea rezecţiei colorectale laparoscopice în cazurile de endometrioză profundă, precum şi ...